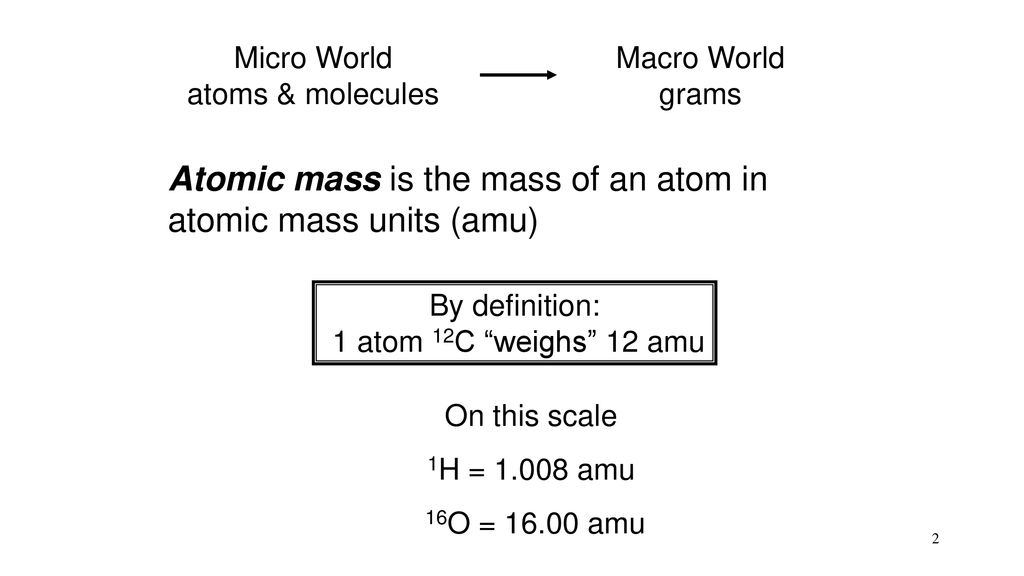
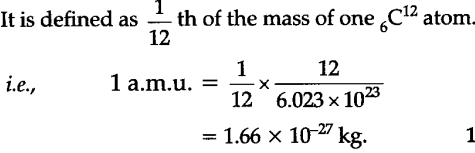Molecular Mass. Mass of Atom Measured in atomic mass unit (amu) 1 amu = 1.66 x g Defined by assigning the mass of 12 amu to the carbon-12 isotope. - ppt download

What is Atomic Mass Unit (amu), its Calculation and Explanation of Average Atomic Mass of Elements - YouTube

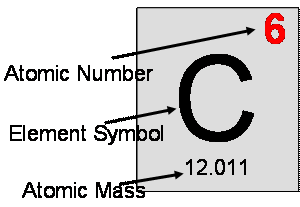
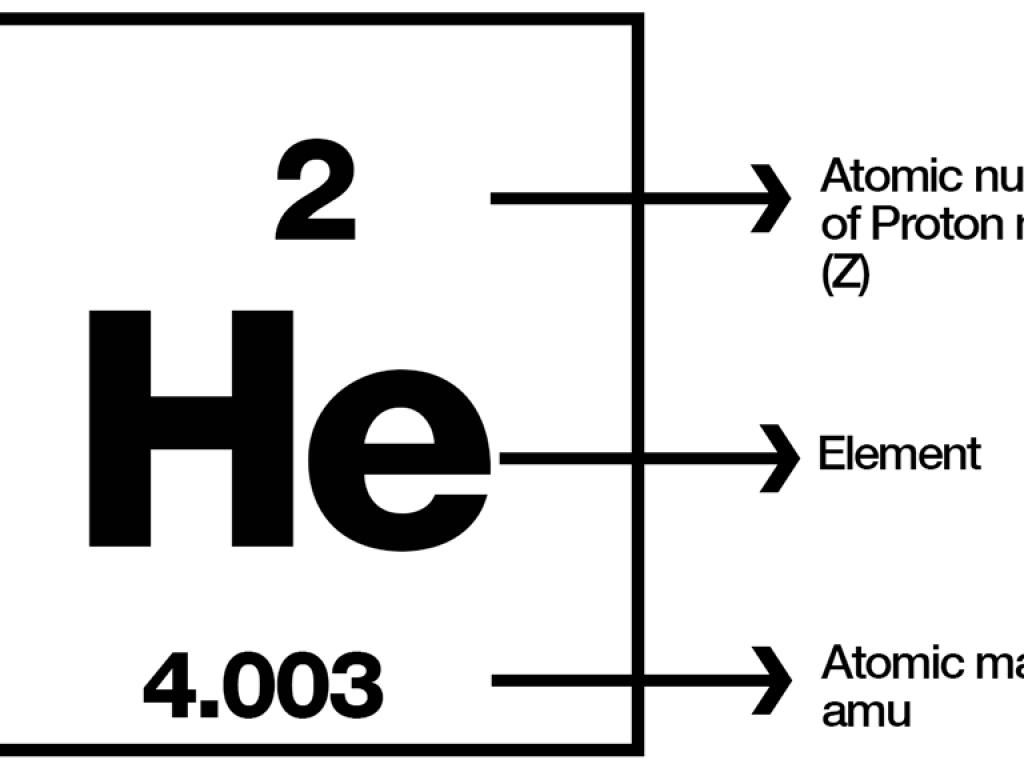
Atomic Weights The mass of an individual atom, ion, or molecule is very small. Scientists use the atomic mass unit (amu) to express the mass of atoms or. - ppt download

Moles Notes. 1. Atomic Mass Unit amu – atomic mass unit, used to describe the mass of an atom Conversion factor: 1 amu = 1.66 x g Equivalence statement: - ppt download