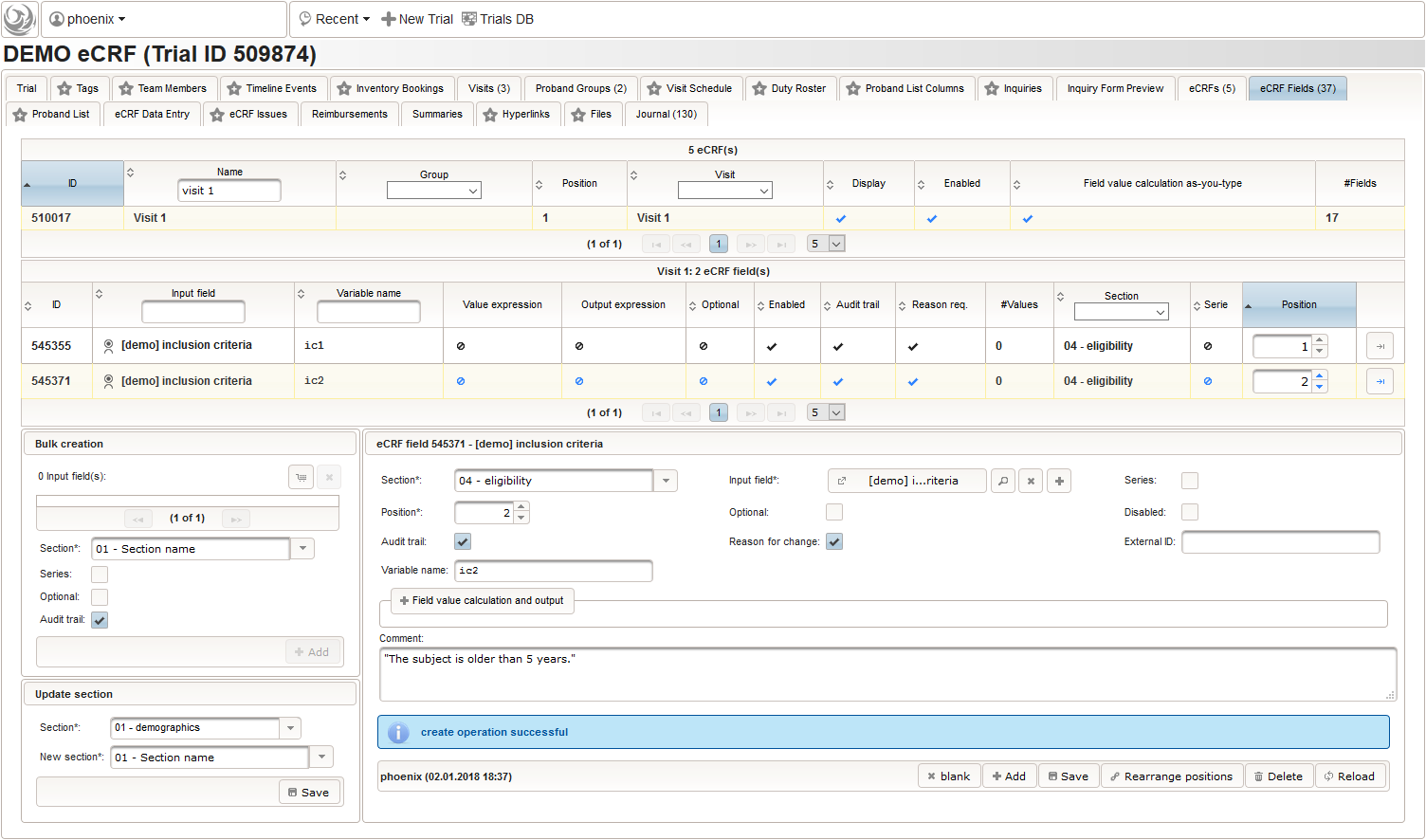
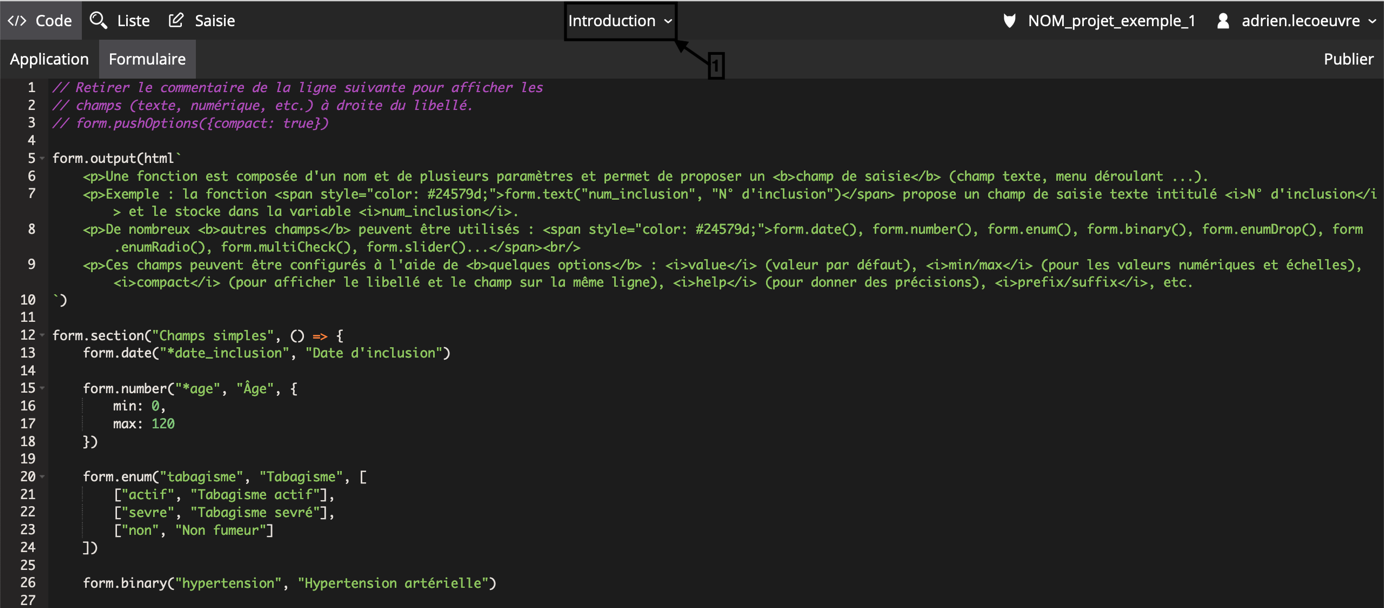
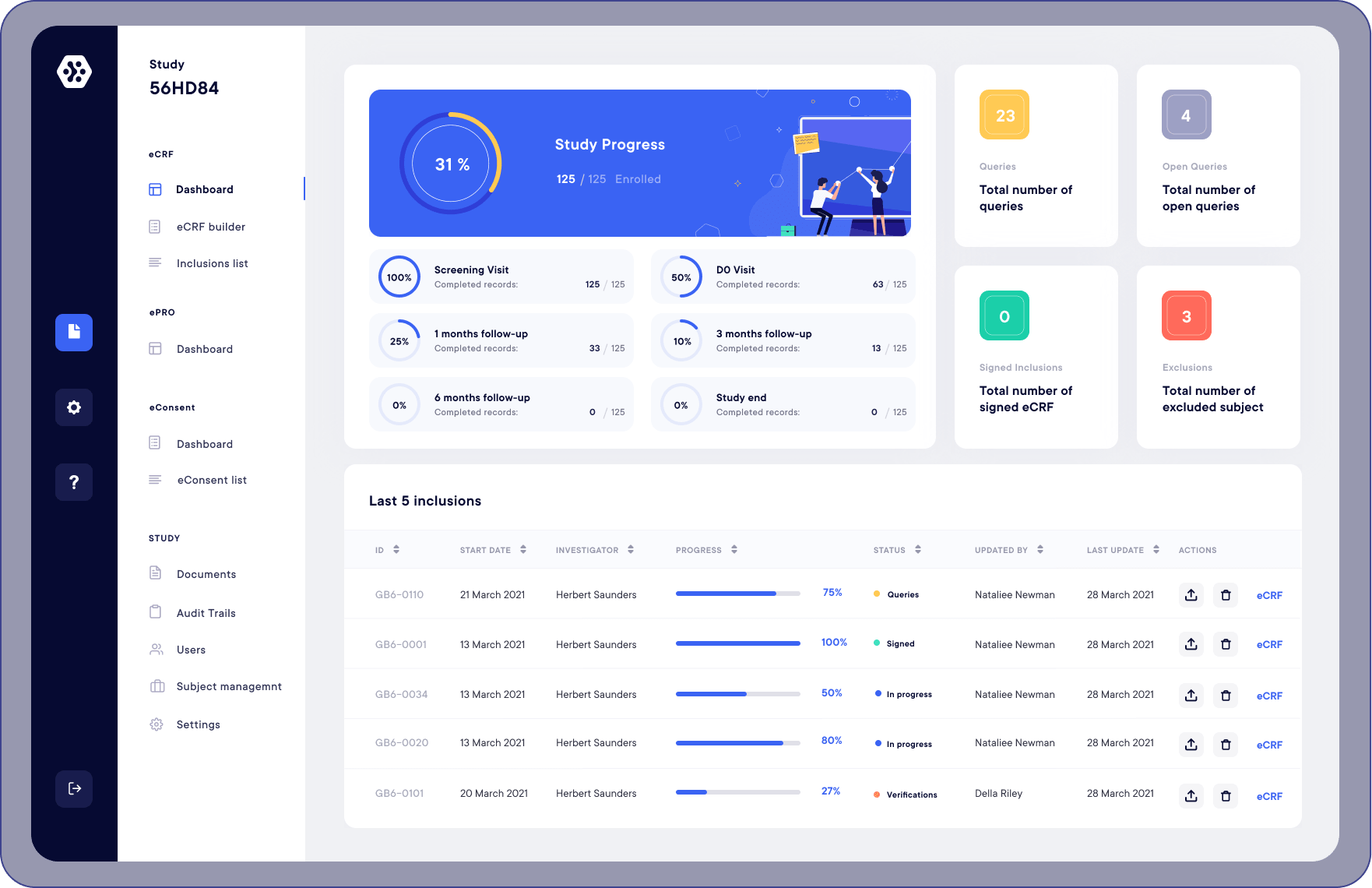
A multi-technique approach to bridge electronic case report form design and data standard adoption - ScienceDirect

PDF) Updating Framingham CVD risk score using waist circumference and estimated cardiopulmonary function: a cohort study based on a southern Xinjiang population

Figure 3 from A pragmatic method for transforming clinical research data from the research electronic data capture "REDCap" to Clinical Data Interchange Standards Consortium (CDISC) Study Data Tabulation Model (SDTM): Development and