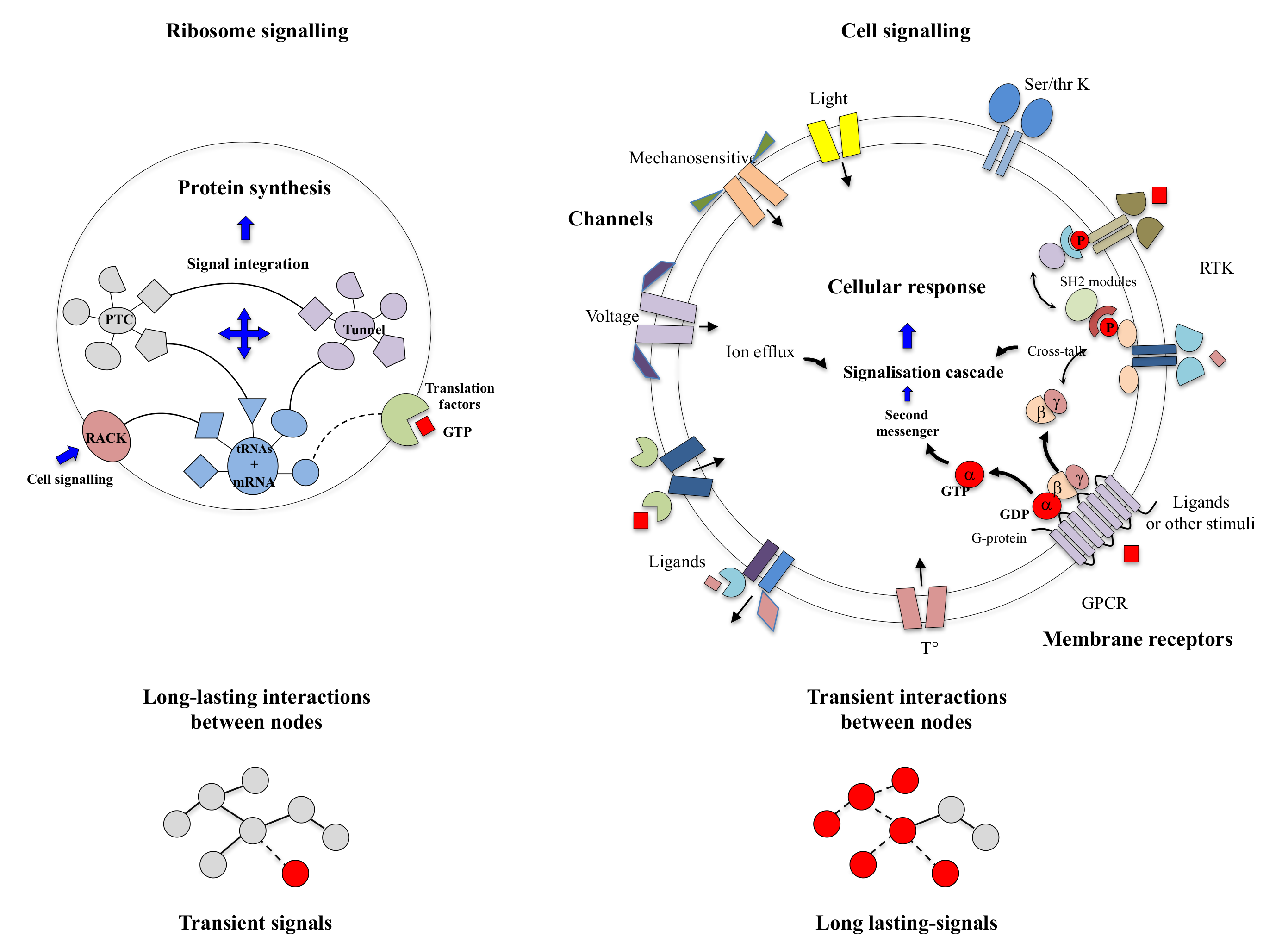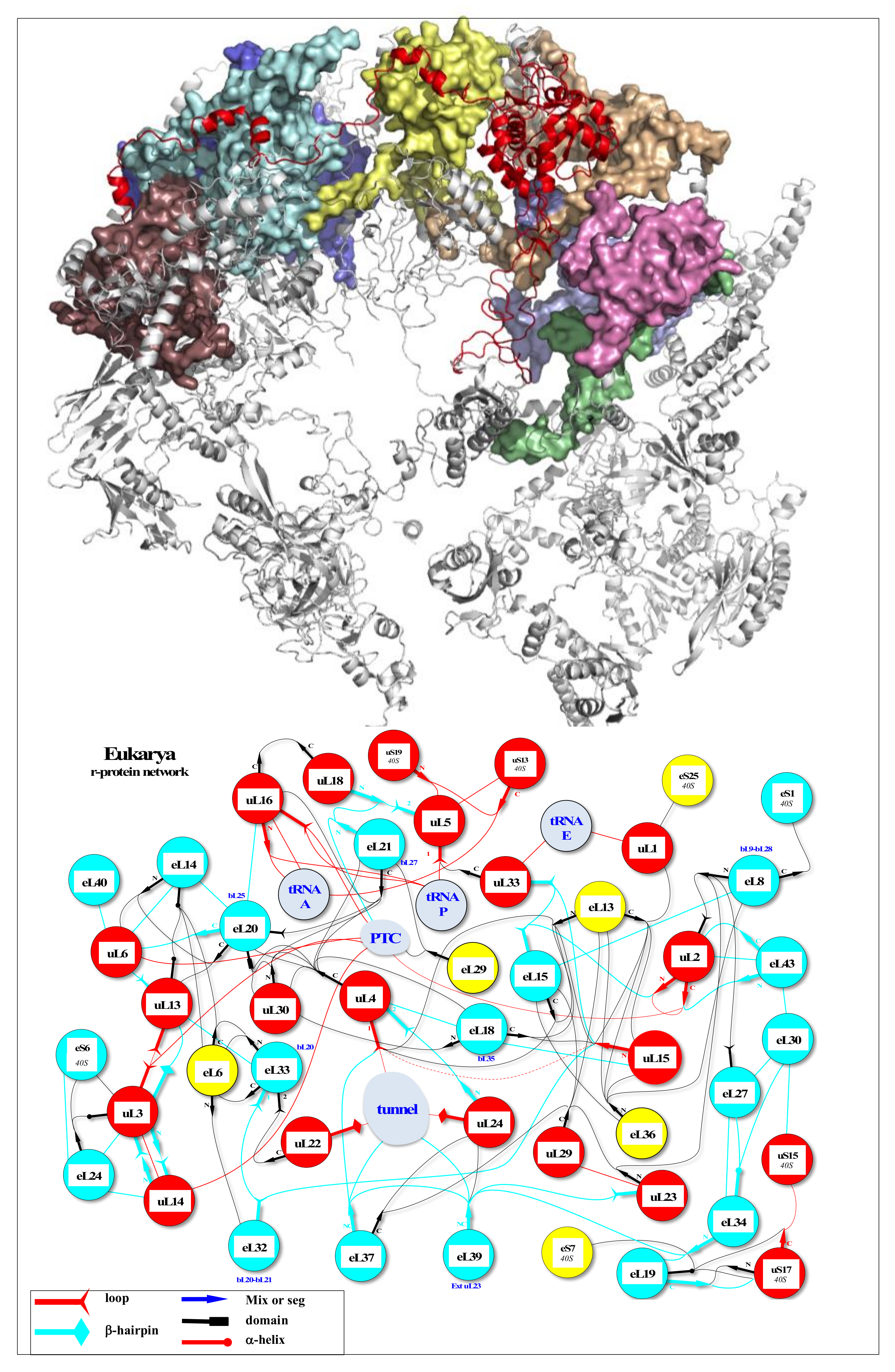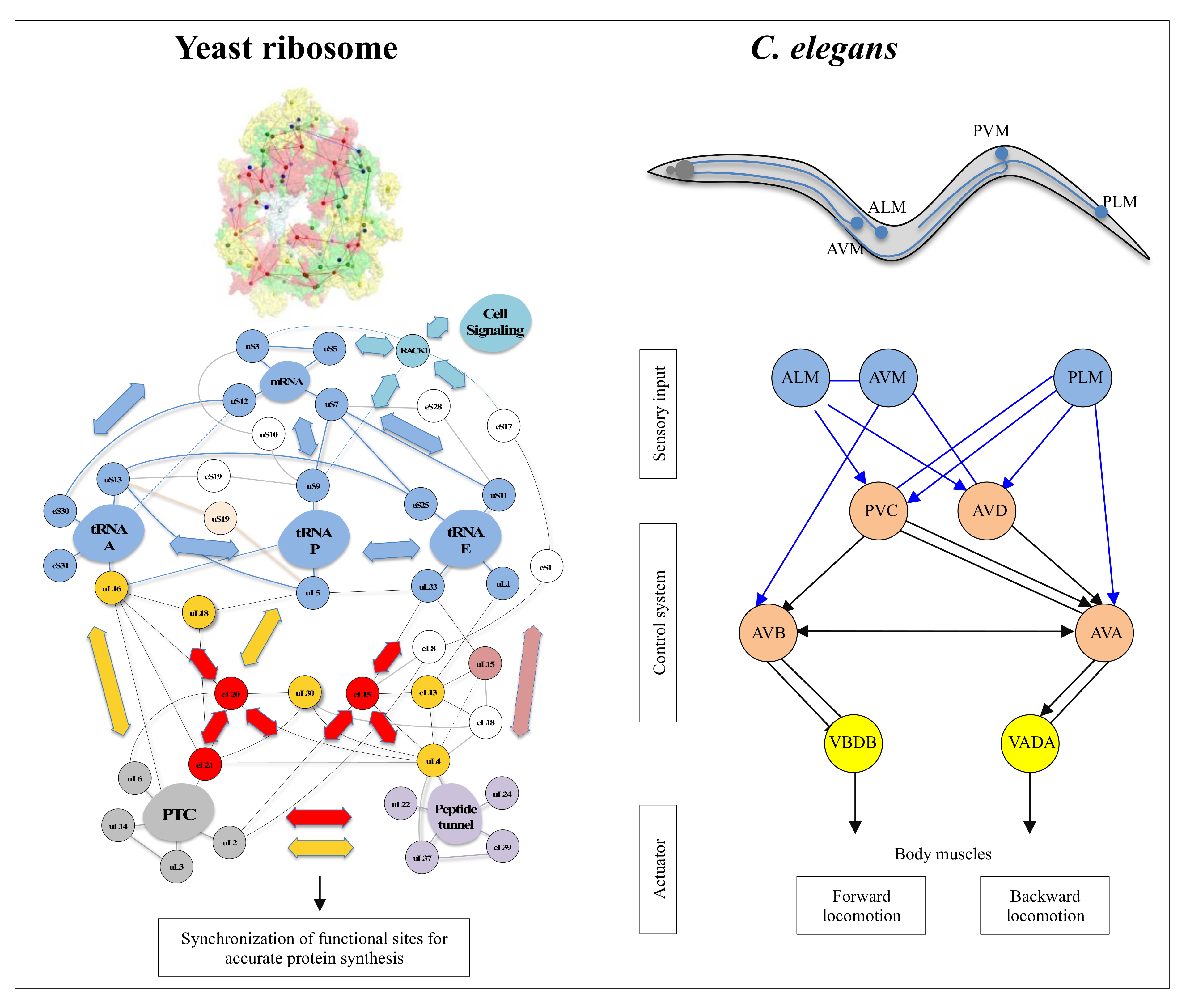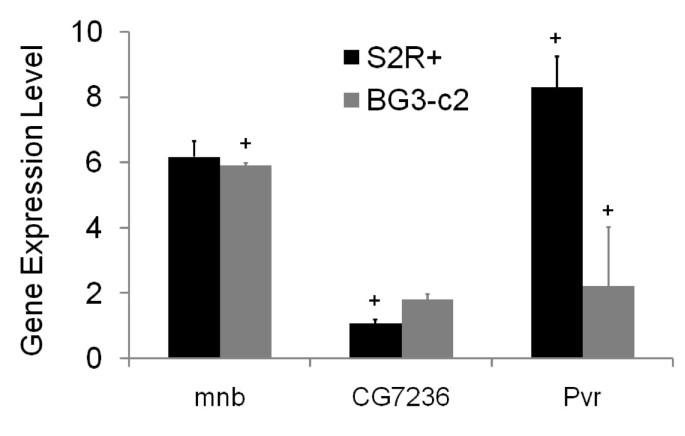
Parallel RNAi screens across different cell lines identify generic and cell type-specific regulators of actin organization and cell morphology | Genome Biology | Full Text

Synthesis, Evaluation, and Mechanism Study of Novel Indole-Chalcone Derivatives Exerting Effective Antitumor Activity Through Microtubule Destabilization in Vitro and in Vivo | Journal of Medicinal Chemistry

Conformation-Based Design and Synthesis of Apratoxin A Mimetics Modified at the α,β-Unsaturated Thiazoline Moiety | Journal of Medicinal Chemistry
![Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0223523421004785-gr4.jpg)
Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect
![Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0223523421004785-gr2.jpg)
Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect

Synthesis, Evaluation, and Mechanism Study of Novel Indole-Chalcone Derivatives Exerting Effective Antitumor Activity Through Microtubule Destabilization in Vitro and in Vivo | Journal of Medicinal Chemistry

PDF) Discovery of a Series of Acridinones as Mechanism-Based Tubulin Assembly Inhibitors with Anticancer Activity
![Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0223523421004785-gr6.jpg)
Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect
![Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0223523421004785-sc2.jpg)
Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect
![Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0223523421004785-ga1.jpg)
Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect

Conformation-Based Design and Synthesis of Apratoxin A Mimetics Modified at the α,β-Unsaturated Thiazoline Moiety | Journal of Medicinal Chemistry
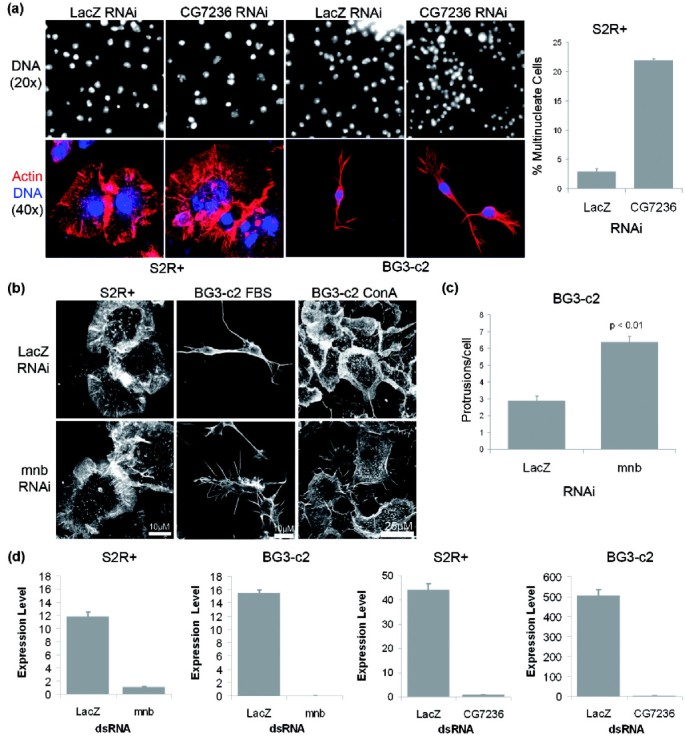
Parallel RNAi screens across different cell lines identify generic and cell type-specific regulators of actin organization and cell morphology | Genome Biology | Full Text
![Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0223523421004785-sc1.jpg)
Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect
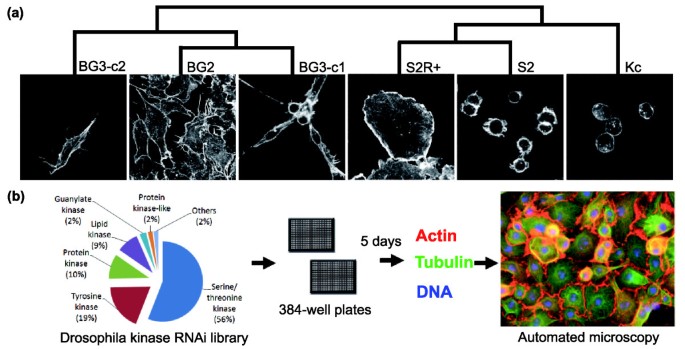
Parallel RNAi screens across different cell lines identify generic and cell type-specific regulators of actin organization and cell morphology | Genome Biology | Full Text
![Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect Design, synthesis and biological evaluation of indole-based [1,2,4]triazolo[4,3-a] pyridine derivatives as novel microtubule polymerization inhibitors - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0223523421004785-gr1.jpg)