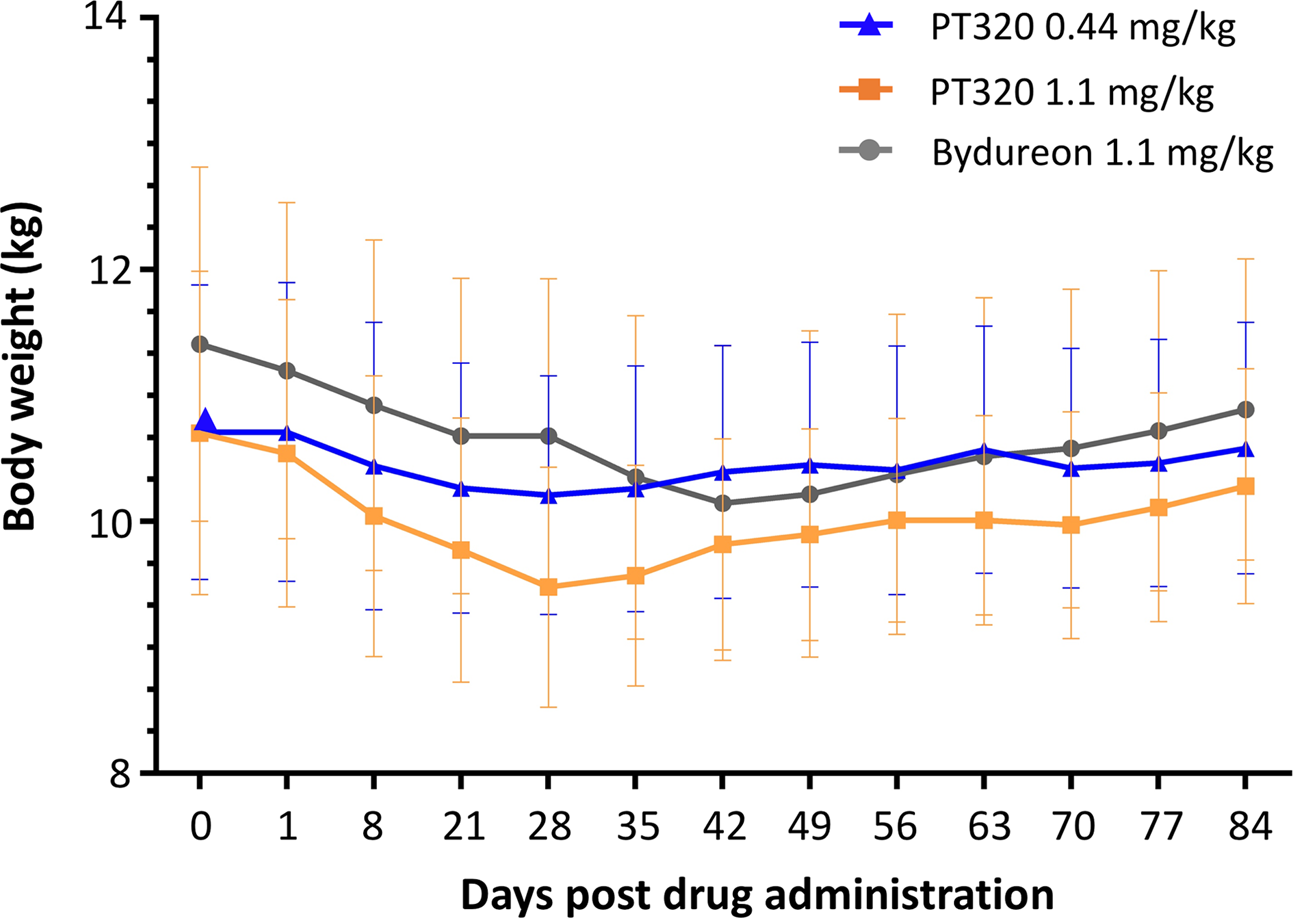
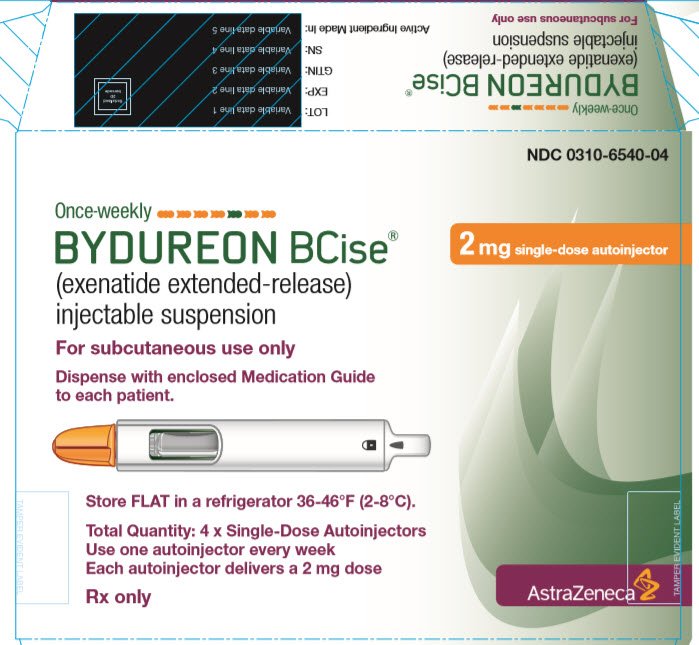
Dosing and Administration | BYDUREON BCise® (exenatide extended-release) injectable suspension 2 mg | For HCPs

Bydureon Bcise (Exenatide Extended-Release Injectable Suspension): Uses, Dosage, Side Effects, Interactions, Warning

AstraZeneca's Bydureon gets a facelift, but can it stand out in a GLP-1 market that's only getting tougher? | Fierce Pharma
Dosing and Administration | BYDUREON BCise® (exenatide extended-release) injectable suspension 2 mg | For HCPs

BYDUREON BCise (exenatide extended-release) Approved in the US for the Treatment of Type 2 Diabetes in Pediatric Patients Ages 10 Years and Older | Business Wire

Bydureon Bcise (Exenatide Extended-Release Injectable Suspension): Uses, Dosage, Side Effects, Interactions, Warning