Adjustment of ηCHx-N intercomponent interaction parameter for nitrogen... | Download Scientific Diagram
Vapor-liquid equilibrium in the system carbon dioxide + n-pentane from 252 to 458 K at pressures to 10 MPa | Journal of Chemical & Engineering Data
Viscosities and Densities of Binary Mixtures of Hexadecane with Dissolved Methane or Carbon Dioxide at Temperatures From (298 to

PVT, saturated liquid density and vapor-pressure measurements of main components of the biofuels at high temperatures and high pressures: Methyl palmitate - ScienceDirect

Experiments of Vapor–Liquid Phase Transition of Fluids Confined in Nanopores: Implications on Modeling | Industrial & Engineering Chemistry Research

Phase behavior, density, and crystallization of polyethylene in n‐pentane and in n‐pentane/CO2 at high pressures - Zhang - 2003 - Journal of Applied Polymer Science - Wiley Online Library

Phase behavior, density, and crystallization of polyethylene in n‐pentane and in n‐pentane/CO2 at high pressures - Zhang - 2003 - Journal of Applied Polymer Science - Wiley Online Library

Vapour-liquid equilibrium of acetone-CO2 mixtures of different compositions at the vicinity of the critical point - ScienceDirect
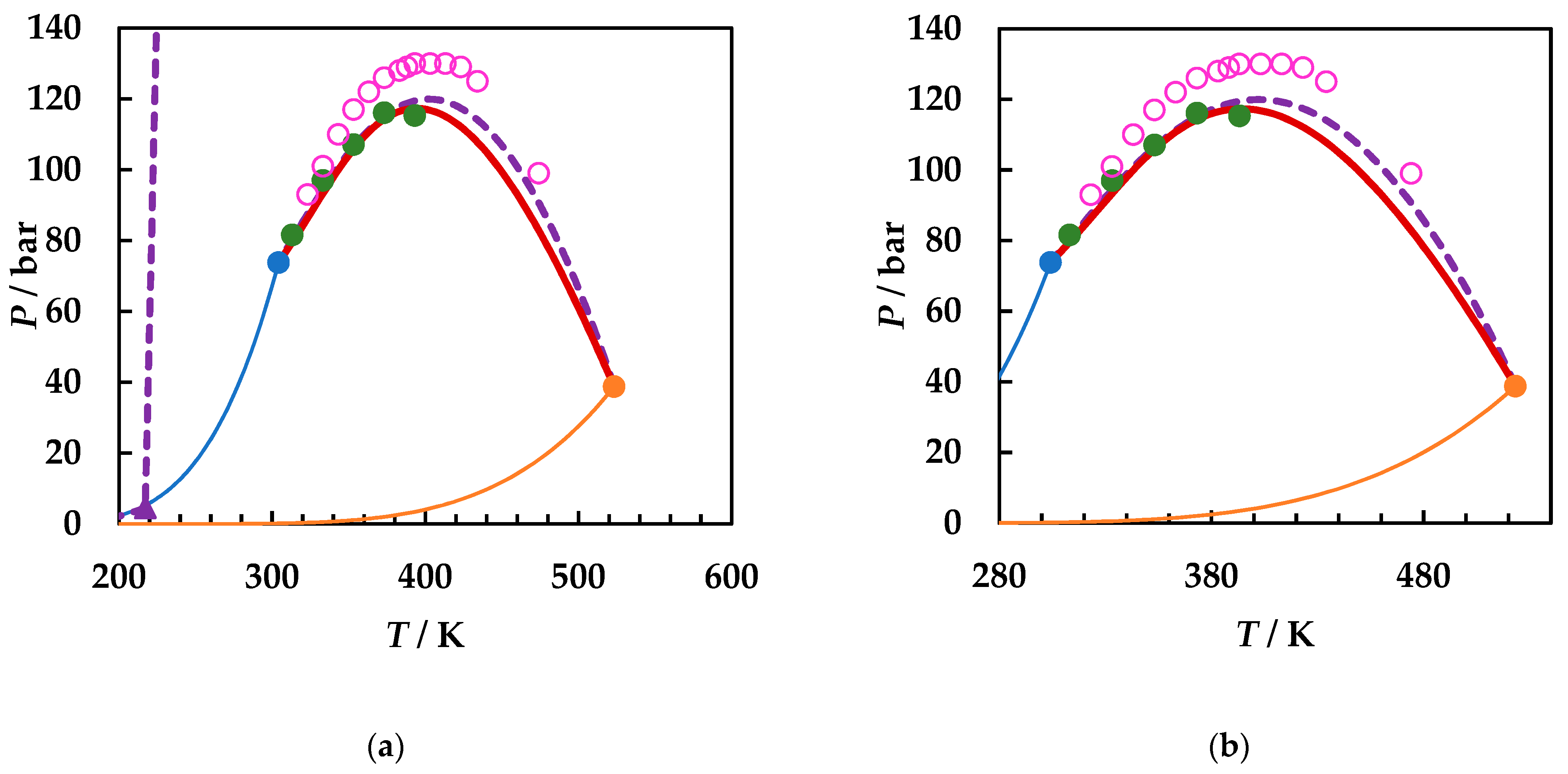
Molecules | Free Full-Text | The Effect of Functional Groups on the Phase Behavior of Carbon Dioxide Binaries and Their Role in CCS | HTML
Experimental pressure versus temperature isochoric – isoplethic curves for n-pentane – dimethyl ether, n-pentane – dimethy

SOLVED: The vapor pressure of liquid pentane, C5H12, is 100. mm Hg at 260 K. A 0.109 g sample of liquid C5H12 is placed in a closed, evacuated 490. mL container at
Correlation of vapor-liquid equilibria for binary mixtures with free energy-based equation of state mixing rules: Carbon dioxide
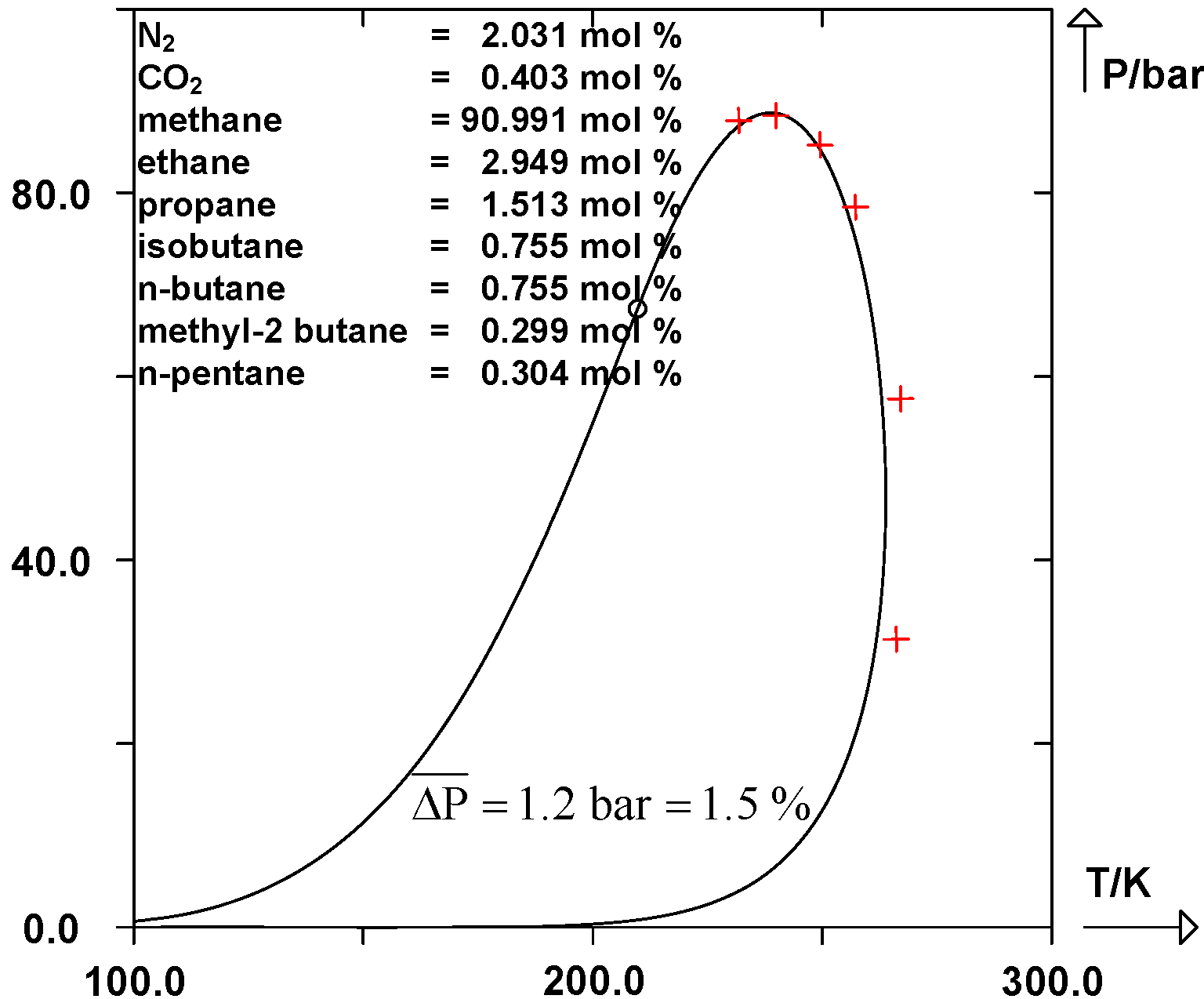
Predicting the Phase Equilibria of Carbon Dioxide Containing Mixtures Involved in CCS Processes Using the PPR78 Model | IntechOpen