Density and Phase Behavior of the CO2 + Methylbenzene System in Wide Ranges of Temperatures and Pressures | Industrial & Engineering Chemistry Research

Density and Phase Behavior of the CO2 + Methylbenzene System in Wide Ranges of Temperatures and Pressures | Industrial & Engineering Chemistry Research

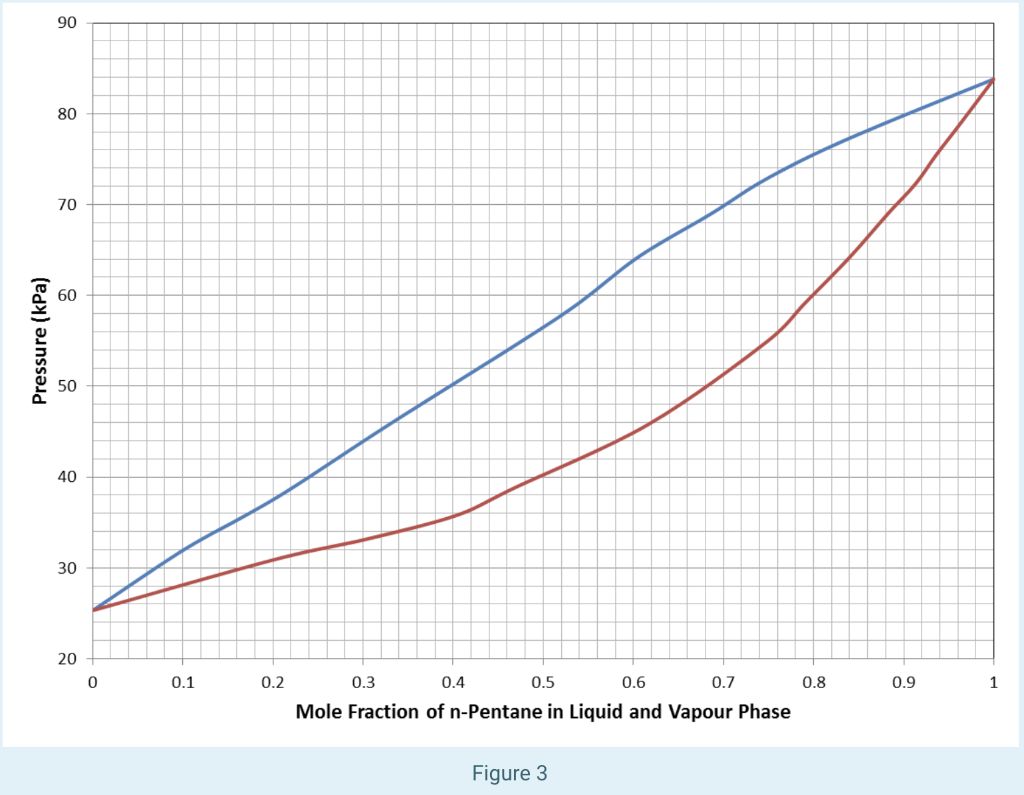
PDF) Design and Operations of Natural Gas Handling Facilities Course prepared for the training department of National Iranian Gas Company | gazi huq - Academia.edu
![PDF] Viscosity of CO2-rich mixtures from 243 K to 423 K at pressures up to 155 MPa: New experimental viscosity data and modelling PDF] Viscosity of CO2-rich mixtures from 243 K to 423 K at pressures up to 155 MPa: New experimental viscosity data and modelling](https://www.researchgate.net/profile/Mahmoud-Nazeri-3/publication/321034612/figure/tbl2/AS:668724877094913@1536447843019/Compositions-of-the-multi-component-mixtures-in-mol-with-the-expanded-uncertainties_Q320.jpg)
PDF] Viscosity of CO2-rich mixtures from 243 K to 423 K at pressures up to 155 MPa: New experimental viscosity data and modelling

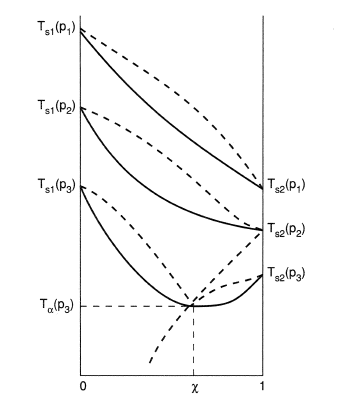
High-Pressure Densities and Excess Molar Volumes for the Binary Mixture of Carbon Dioxide and Hydrogen Sulfide at T = 343–397 K | Journal of Chemical & Engineering Data

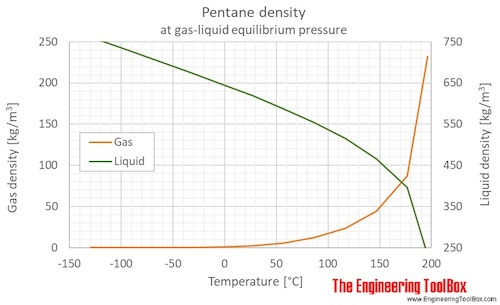
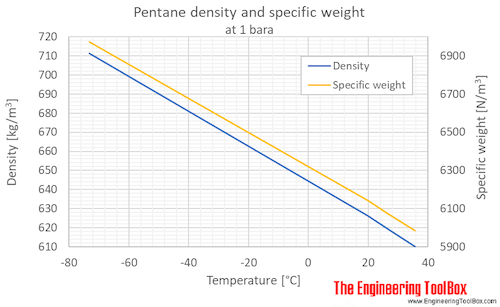
New compact Equations for the Compressibility Factor Z and Density of Liquid and Vapor Carbon Dioxide | Chem-Eng-Musings

High-temperature and high-pressure density and liquid–vapor phase transition properties of methyl octanoate as main biofuel component - ScienceDirect
![PDF] Vapor–Liquid Equilibrium Data for the Carbon Dioxide (CO2) + 1,1,1,3,3-Pentafluorobutane (R365mfc) System at Temperatures from 283.15 to 337.15 K | Semantic Scholar PDF] Vapor–Liquid Equilibrium Data for the Carbon Dioxide (CO2) + 1,1,1,3,3-Pentafluorobutane (R365mfc) System at Temperatures from 283.15 to 337.15 K | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/41236103d2b9c81bab8dfe341d7be95b4e6fb18d/21-Figure1-1.png)
PDF] Vapor–Liquid Equilibrium Data for the Carbon Dioxide (CO2) + 1,1,1,3,3-Pentafluorobutane (R365mfc) System at Temperatures from 283.15 to 337.15 K | Semantic Scholar

Density and Phase Behavior of the CO2 + Methylbenzene System in Wide Ranges of Temperatures and Pressures | Industrial & Engineering Chemistry Research
![PDF] Viscosity of CO2-rich mixtures from 243 K to 423 K at pressures up to 155 MPa: New experimental viscosity data and modelling PDF] Viscosity of CO2-rich mixtures from 243 K to 423 K at pressures up to 155 MPa: New experimental viscosity data and modelling](https://www.researchgate.net/profile/Mahmoud-Nazeri-3/publication/321034612/figure/tbl3/AS:668724877082624@1536447843050/Parameters-in-the-LBC-and-CO2-LBC-Viscosity-Correlation_Q320.jpg)
PDF] Viscosity of CO2-rich mixtures from 243 K to 423 K at pressures up to 155 MPa: New experimental viscosity data and modelling

Density and Phase Behavior of the CO2 + Methylbenzene System in Wide Ranges of Temperatures and Pressures | Industrial & Engineering Chemistry Research

Percentage deviations of various data sets for saturated densities of... | Download Scientific Diagram