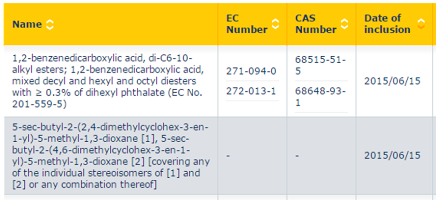
EC number:258-649-2 1. In vitro gene mutation study in bacteria (Annex VII, Section 8.4.1.; test method: Bacterial reverse mutat

SciELO - Saúde Pública - Information gathering for CLP classification Information gathering for CLP classification

Guidance for the identification of endocrine disruptors in the context of Regulations (EU) No 528/2012 and (EC) No 1107/2009 - - 2018 - EFSA Journal - Wiley Online Library
Explanatory note to the proposed guideline on the scope of Annex XVII Entry 63: Lead and its compounds in articles intended for
Dear Valued Customers, We hereby certify that Navitar Inc. fully complies with the related requirements of European Union Regula
![PDF] A comparison of mandatory and voluntary approaches to the implementation of Globally Harmonized System of Classification and Labelling of Chemicals (GHS) in the management of hazardous chemicals. | Semantic Scholar PDF] A comparison of mandatory and voluntary approaches to the implementation of Globally Harmonized System of Classification and Labelling of Chemicals (GHS) in the management of hazardous chemicals. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/887664a93d3aef9a75f12a6dac82c7efa4ddaf9f/4-Table1-1.png)


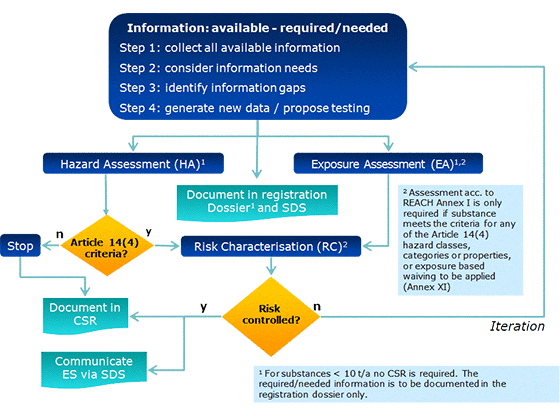


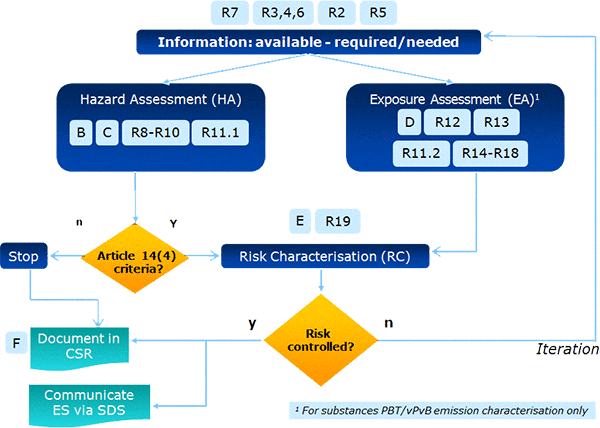
![ECHA - Guidance of the application of CLP criteria [November 2012] ECHA - Guidance of the application of CLP criteria [November 2012]](https://img.yumpu.com/41418795/1/500x640/echa-guidance-of-the-application-of-clp-criteria-november-2012.jpg)