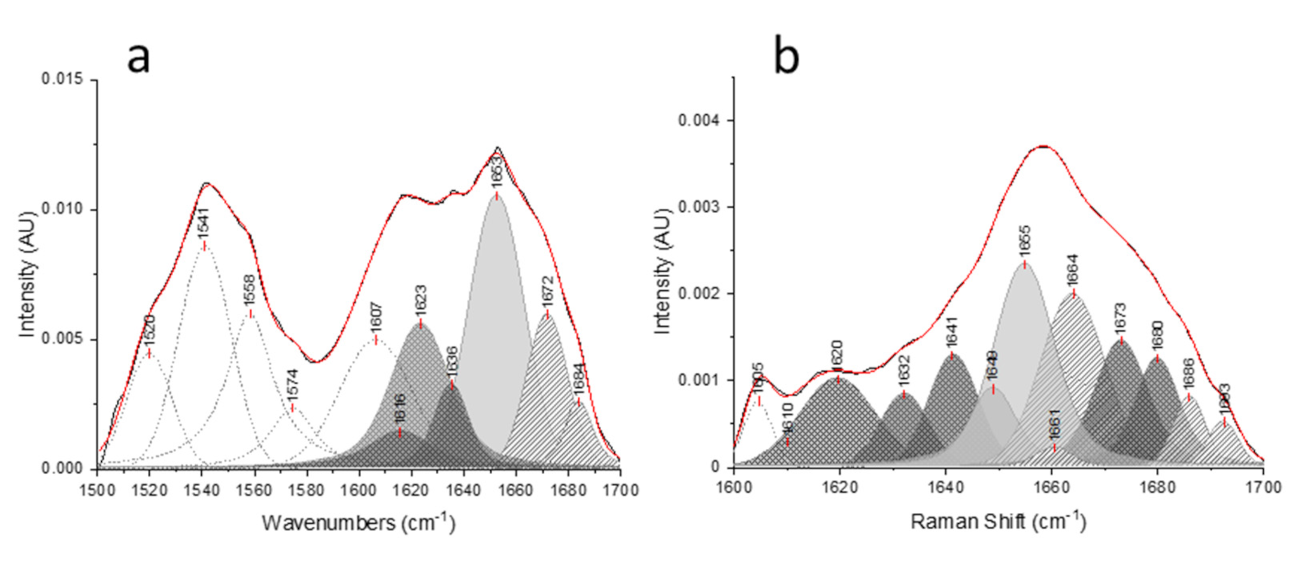
Applied Sciences | Free Full-Text | Peak Fitting Applied to Fourier Transform Infrared and Raman Spectroscopic Analysis of Proteins

Figure 2 from Curve-fitting micro-ATR-FTIR studies of the amide I and II bands of type I collagen in archaeological bone materials | Semantic Scholar

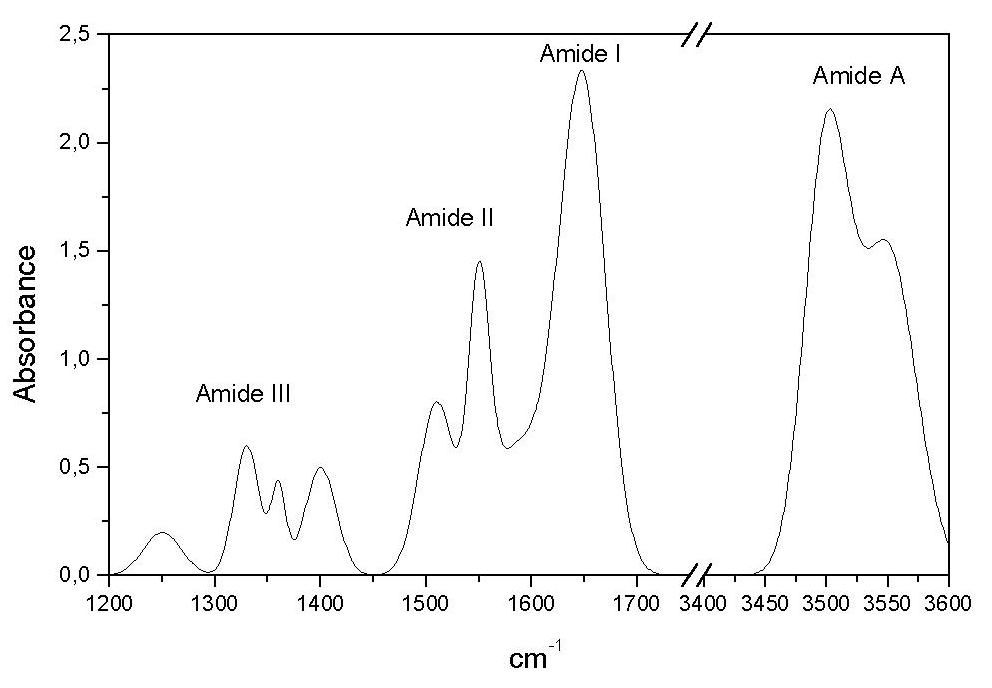
FTIR investigation of the secondary structure of type I collagen: New insight into the amide III band - ScienceDirect

DFT-Calculated IR Spectrum Amide I, II, and III Band Contributions of N-Methylacetamide Fine Components | ACS Omega
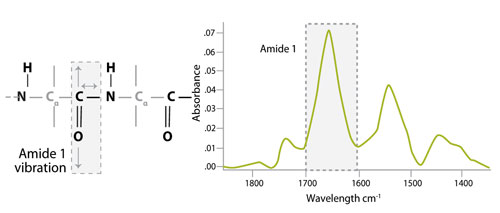
ATR-FTIR spectra showing the Amide I, Amide II, and Amide III regions.... | Download Scientific Diagram

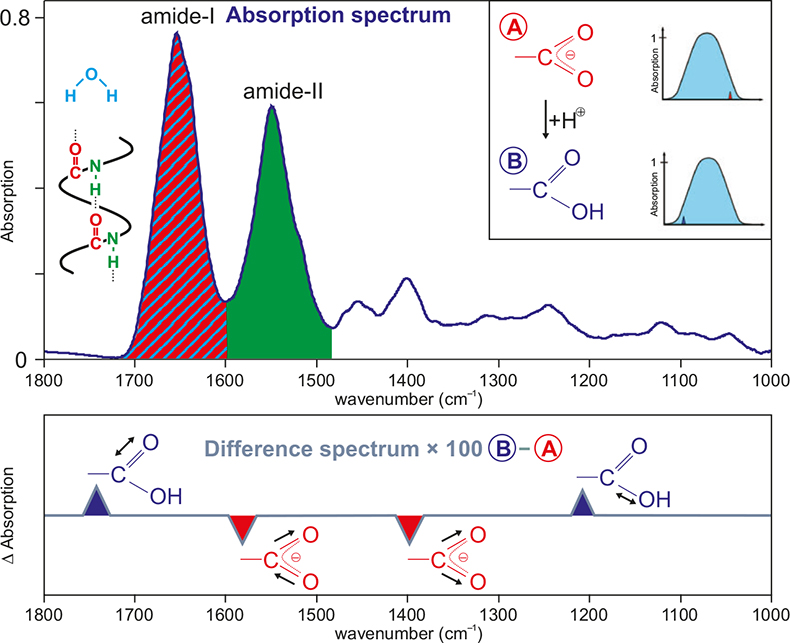
Saturation effects in FTIR spectroscopy: Intensity of amide I and amide II bands in protein spectra | Semantic Scholar

FTIR spectral profiles of Amide I and Amide II bands corresponding to native H3 histones and 3-DG-glycated H3 histone.

DFT-Calculated IR Spectrum Amide I, II, and III Band Contributions of N-Methylacetamide Fine Components | ACS Omega

DFT-Calculated IR Spectrum Amide I, II, and III Band Contributions of N-Methylacetamide Fine Components | ACS Omega
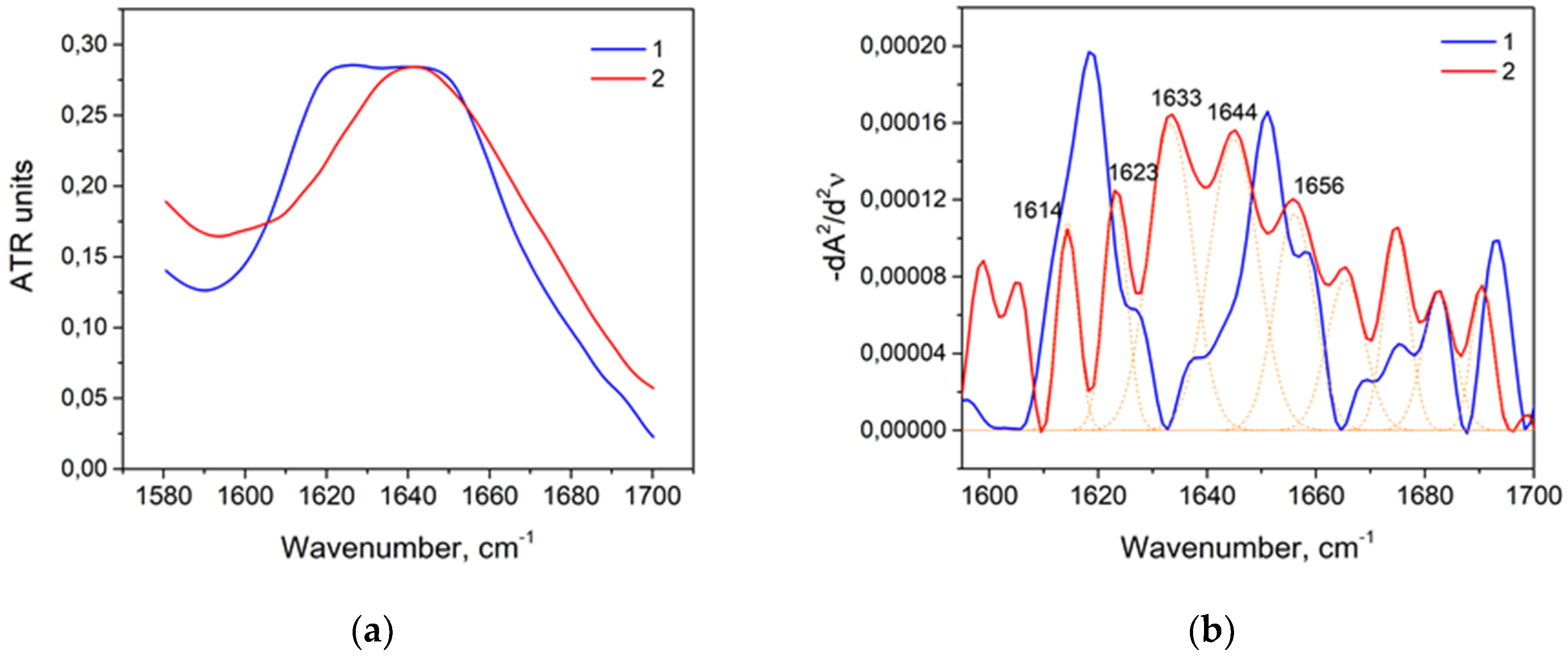
Biomolecules | Free Full-Text | Systematic FTIR Spectroscopy Study of the Secondary Structure Changes in Human Serum Albumin under Various Denaturation Conditions
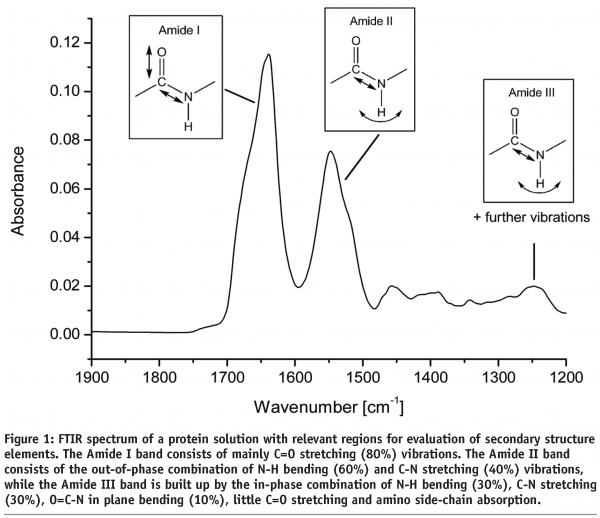
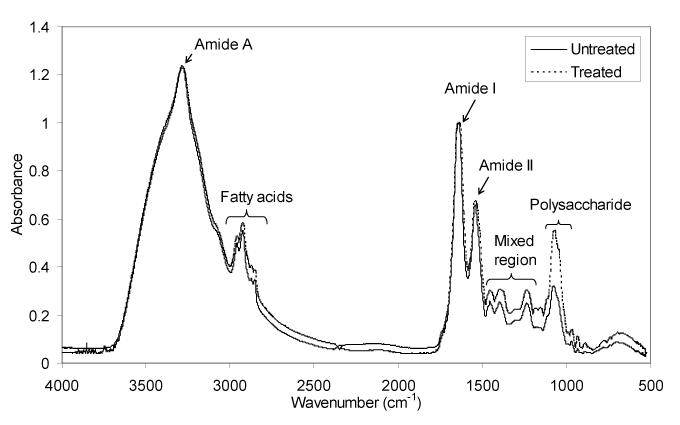
The determination of structural changes of biopharmaceuticals during Freeze-Drying using Fourier Transform Infrared Spectroscopyb - European Pharmaceutical Review
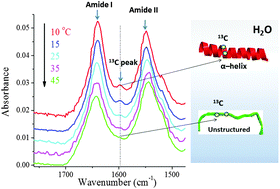
The 13C amide I band is still sensitive to conformation change when the regular amide I band cannot be distinguished at the typical position in H2O - Chemical Communications (RSC Publishing)