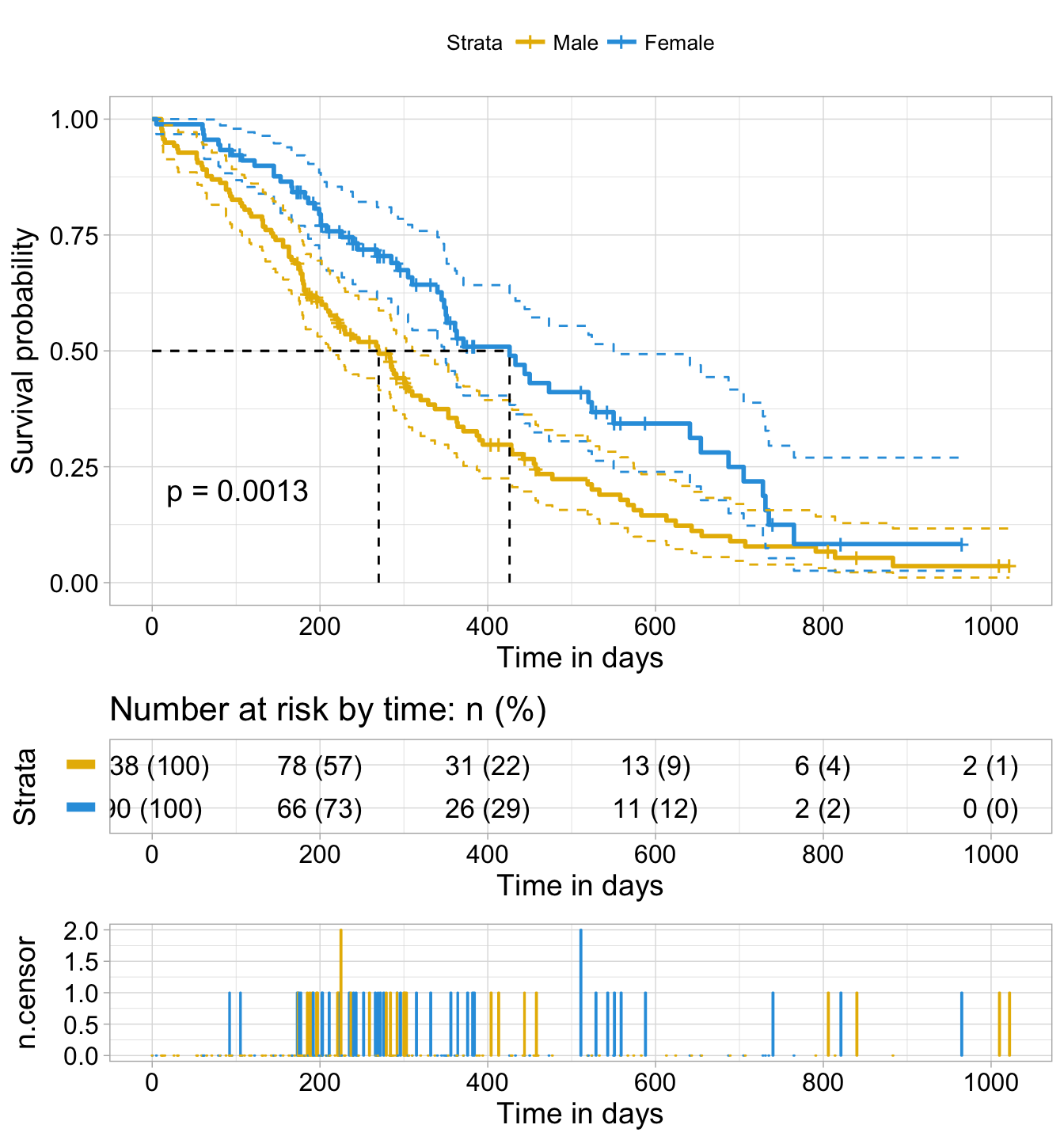
Progression-free survival by IRC Shown is the Kaplan-Meier curve for... | Download Scientific Diagram

Recurrence-free survival versus overall survival as a primary endpoint for studies of resected colorectal liver metastasis: a retrospective study and meta-analysis - The Lancet Oncology

The validity of progression‐free survival 2 as a surrogate trial end point for overall survival - Woodford - 2022 - Cancer - Wiley Online Library

Progression-free survival (PFS) curve and overall survival (OS) curve... | Download Scientific Diagram

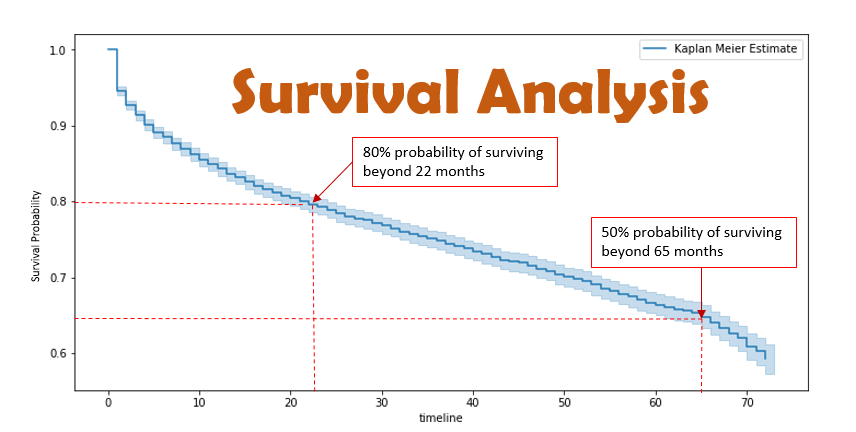
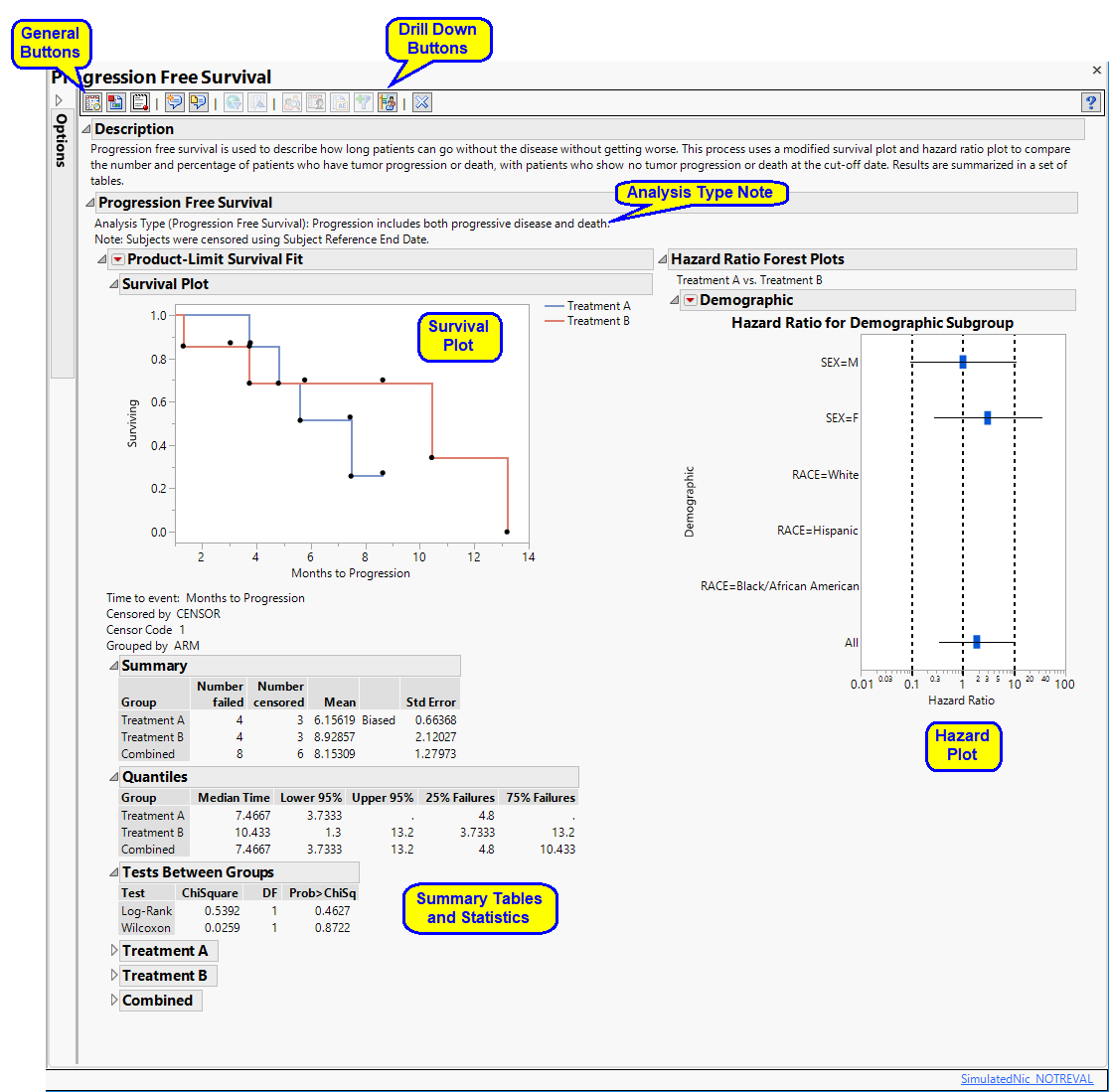
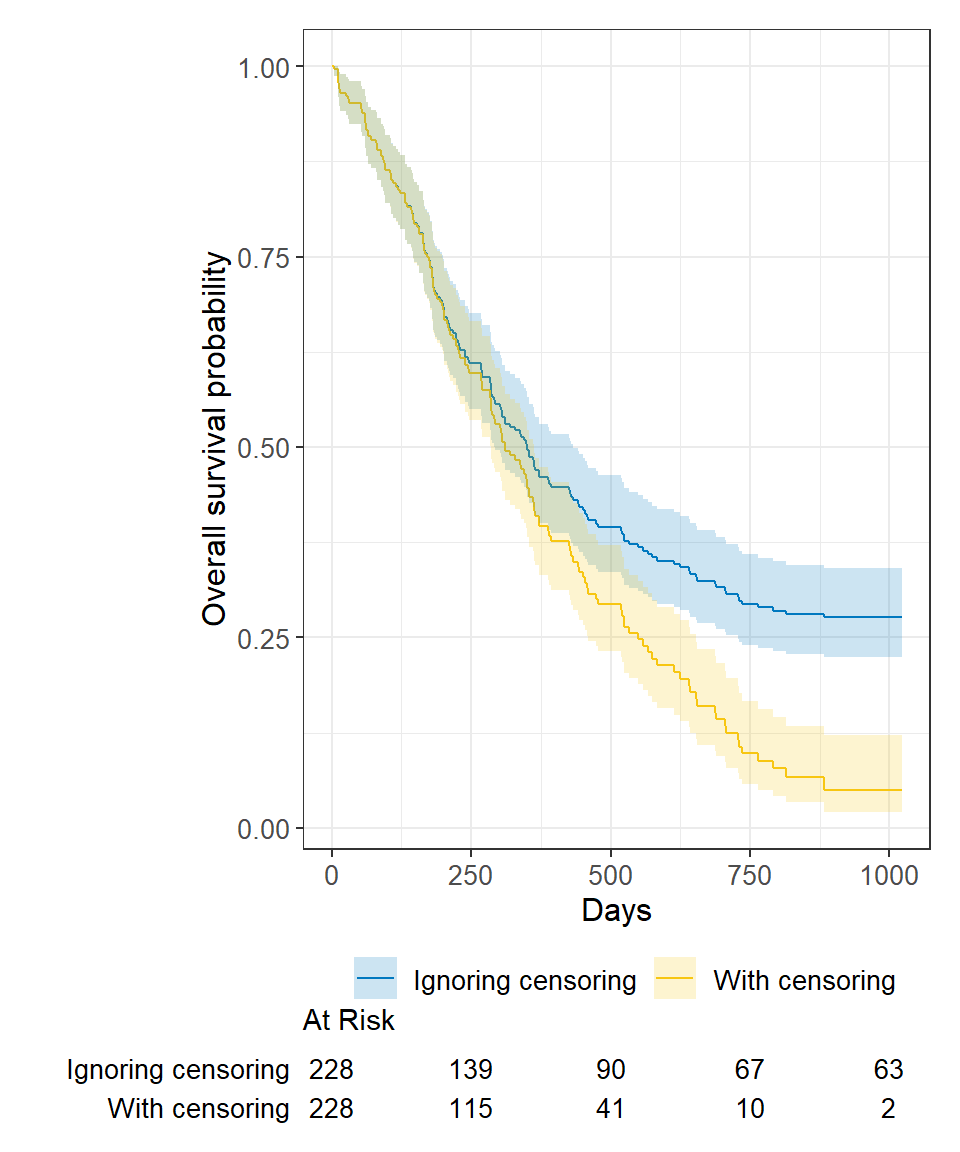
On Biostatistics and Clinical Trials: Understanding the endpoints in oncology: overall survival, progression free survival, hazard ratio, censored value

Progression-free survival and overall survival curves for all patients... | Download Scientific Diagram
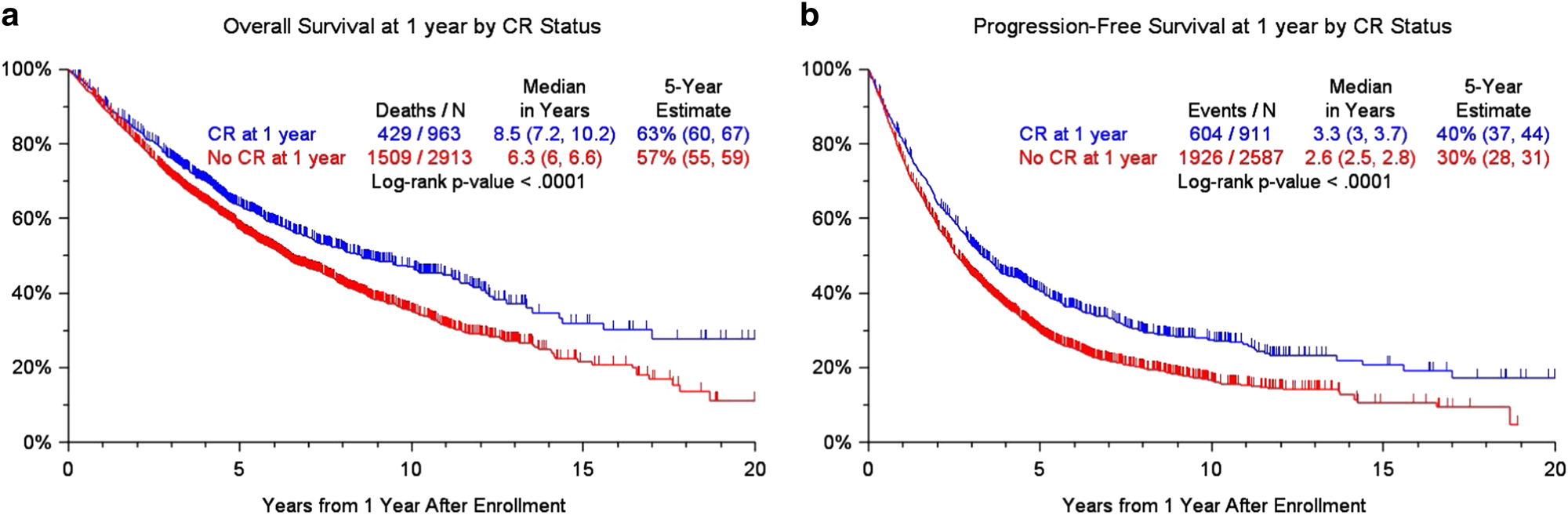
Clinical predictors of long-term survival in newly diagnosed transplant eligible multiple myeloma — an IMWG Research Project | Blood Cancer Journal
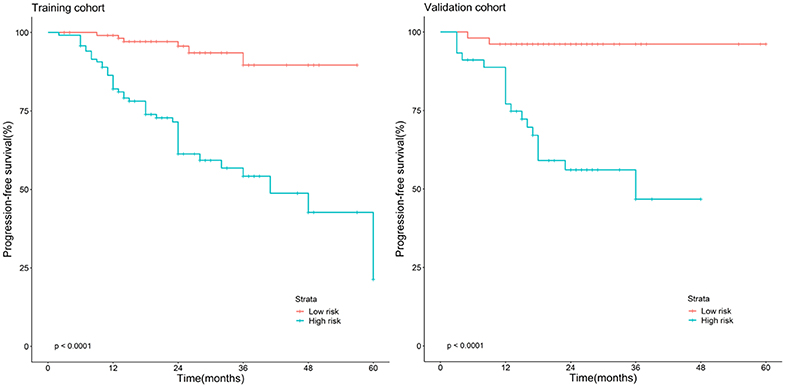
Frontiers | Predicting Progression-Free Survival Using MRI-Based Radiomics for Patients With Nonmetastatic Nasopharyngeal Carcinoma

Progression-free survival at 24 months and subsequent survival of patients with extranodal NK/T-cell lymphoma: a China Lymphoma Collaborative Group (CLCG) study | Leukemia
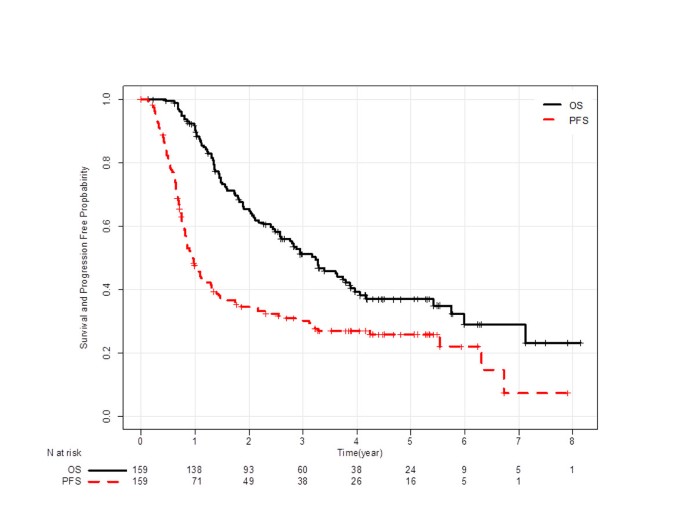
Progression-free survival at 2 years is a reliable surrogate marker for the 5-year survival rate in patients with locally advanced non-small cell lung cancer treated with chemoradiotherapy | BMC Cancer | Full

Progression-free survival (PFS) curve and overall survival (OS) curve... | Download Scientific Diagram