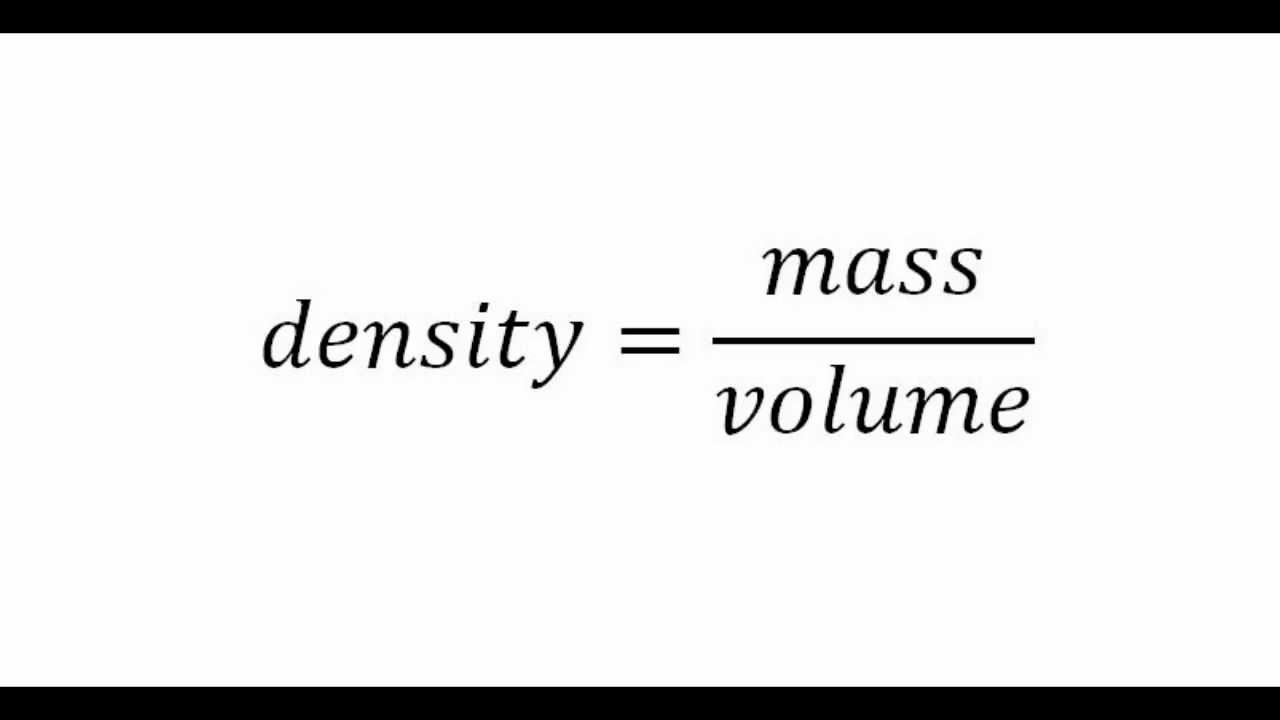Calculate the atmospheric pressuremeasured by the barometer for agiven density of mercury as - Brainly.in

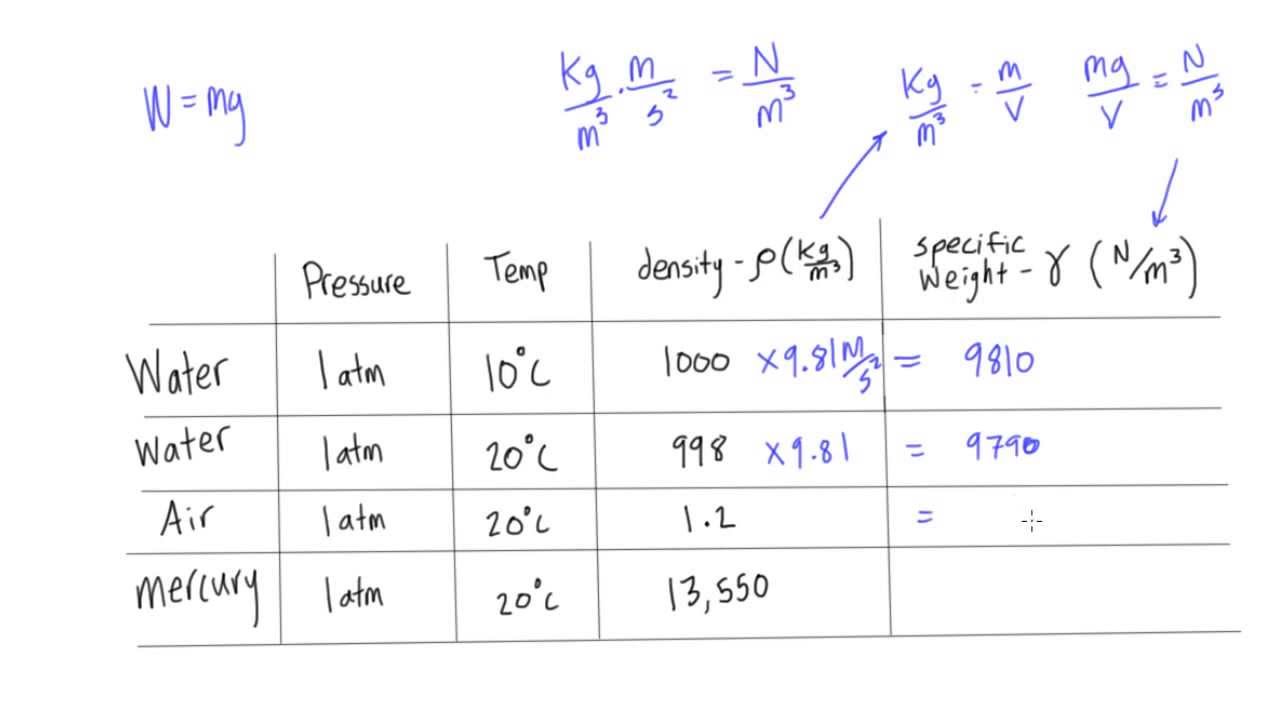
The height of mercury column in a barometer in a Calcutta laboratory was recorded to be 75 cm. Calculate this pressure in SI and CGS units the following data, Specific gravity of

The density of liquid mercury is `13.6 g//cm^(3).` How many moles of mercury are there in 1 - YouTube
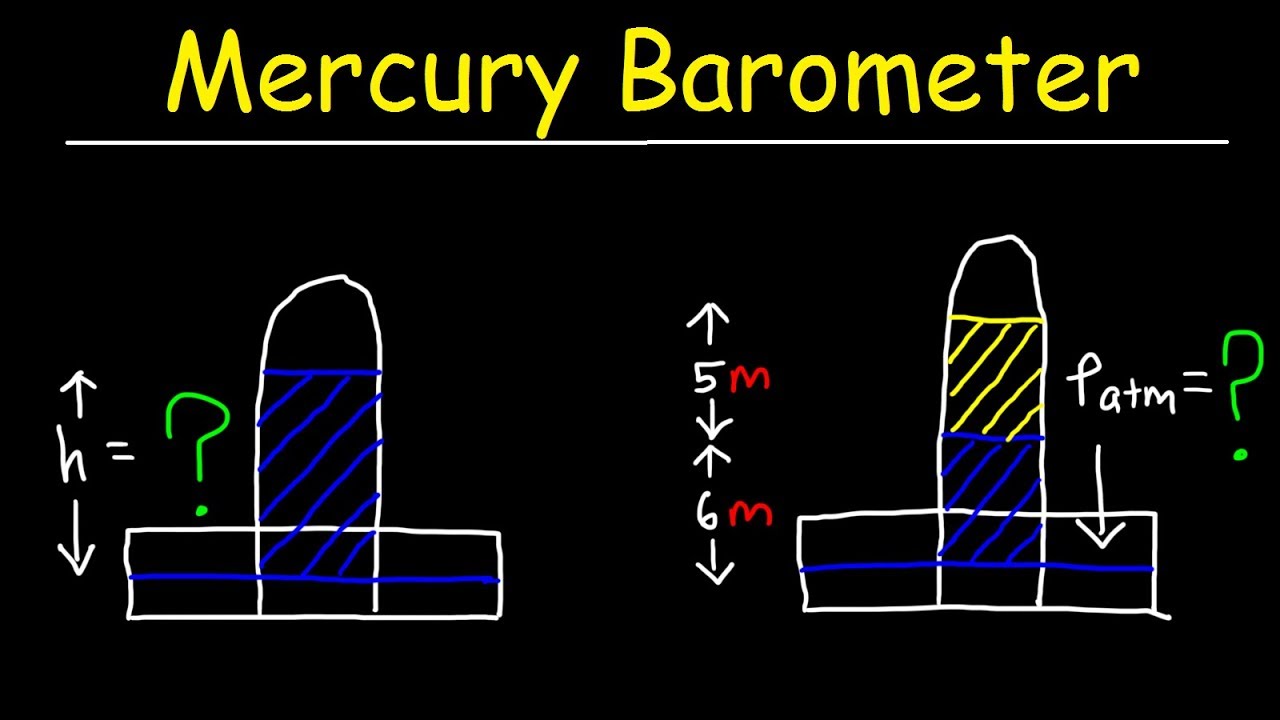
Mercury Barometer Problems, Physics - Air Pressure, Height & Density Calculations - Fluid Statics - YouTube

Calculate the mass of 1 litre of moist air at 27^(@)Cwhen the barometer reads 753.6mm of mercury and the dew point is 16.1^(2)C. Saturation vapour pressure of water at 16.1^(@)=13.6mmof mercury, density
The density of mercury is 13.6 g/mL. Calculate approximately the diameter of an atom of mercury - Sarthaks eConnect | Largest Online Education Community
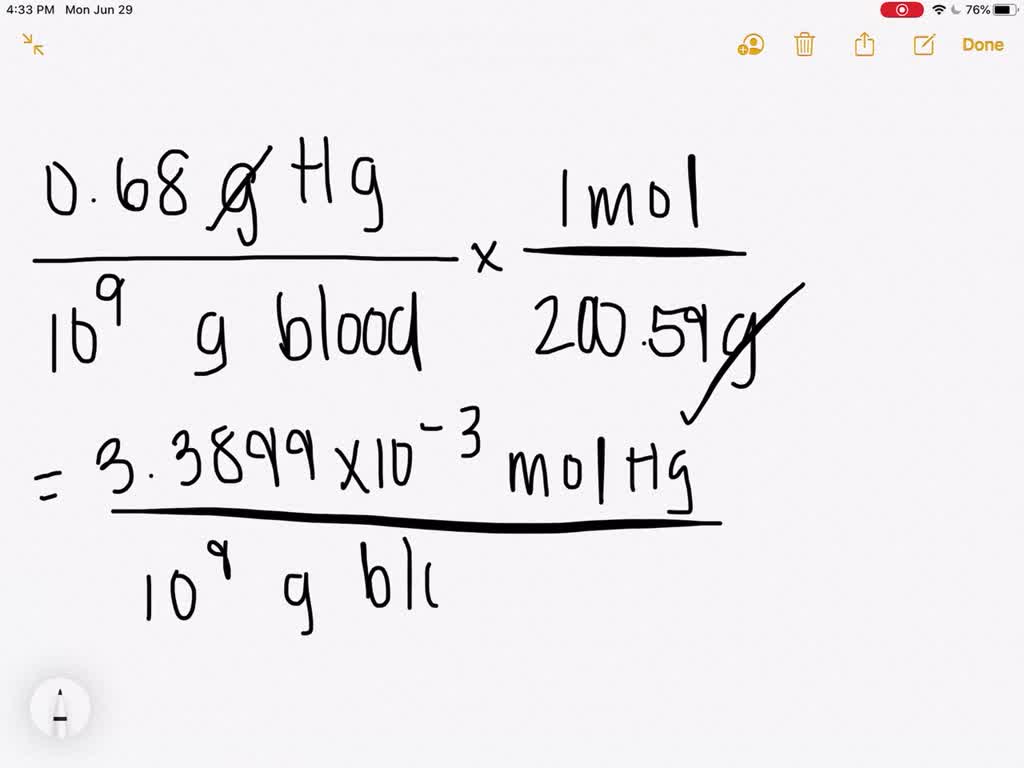


![Mercury Element (Hg) [Liquid Metal] - Density, Boiling Point, Properties & Uses of Mercury Element Mercury Element (Hg) [Liquid Metal] - Density, Boiling Point, Properties & Uses of Mercury Element](https://cdn1.byjus.com/wp-content/uploads/2018/11/chemistry/wp-content/uploads/2016/07/1-1.png)