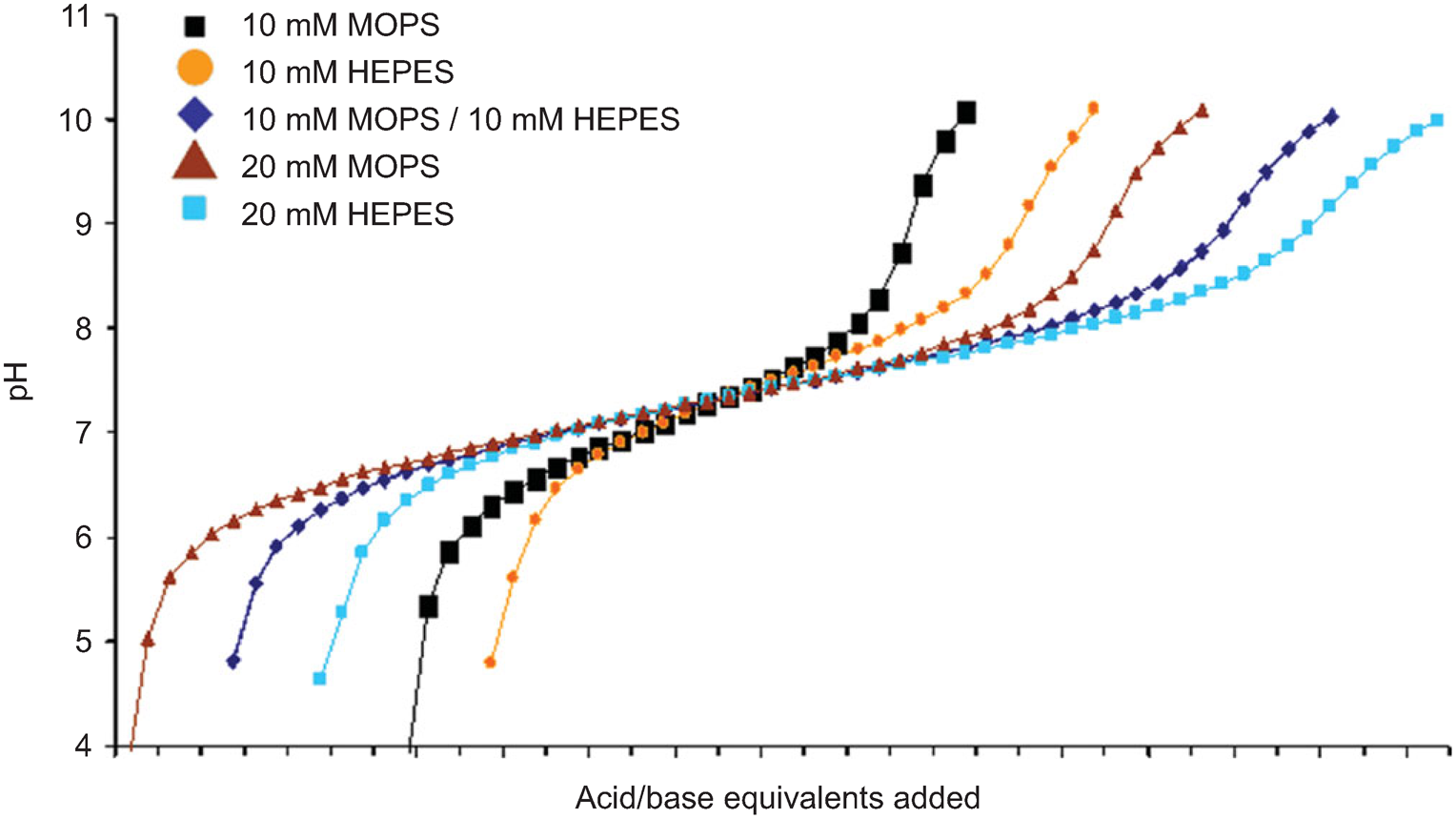
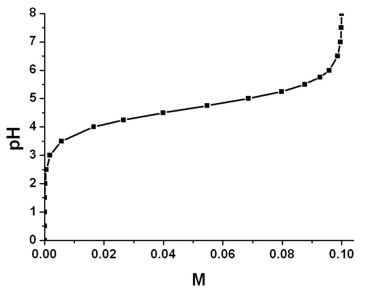
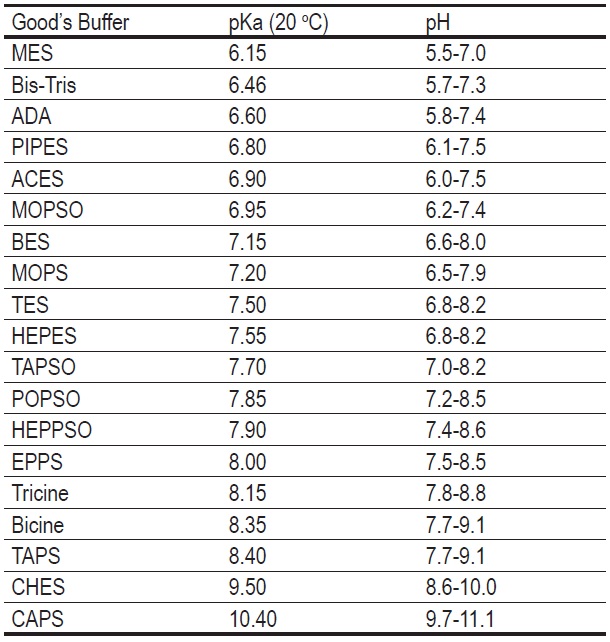
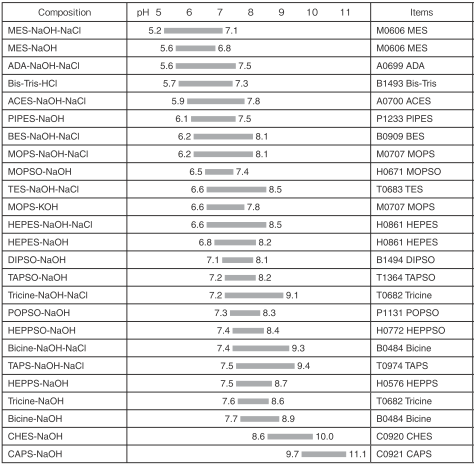
Effect of Buffer on Protein Stability in Aqueous Solutions: A Simple Protein Aggregation Model | The Journal of Physical Chemistry B
Chemistry | Free Full-Text | Appropriate Buffers for Studying the Bioinorganic Chemistry of Silver(I)

Table 4 from Buffer Standards for the Biochemical pH of the Zwitterionic Buffer N-Tris-(Hydroxymethyl) Methyl-2-Aminoethanesulfonic Acid (TES) from 5℃ to 55℃ | Semantic Scholar

Figure 1 from (Un)suitability of the use of pH buffers in biological, biochemical and environmental studies and their interaction with metal ions – a review | Semantic Scholar
![Stability of the Glu[−] sensor (20 µM) in MOPS buffer (20 mM, pH 7.3)... | Download Scientific Diagram Stability of the Glu[−] sensor (20 µM) in MOPS buffer (20 mM, pH 7.3)... | Download Scientific Diagram](https://www.researchgate.net/publication/335330768/figure/fig1/AS:962853695651845@1606573618237/Stability-of-the-Glu-sensor-20M-in-MOPS-buffer-20mM-pH-73-at-25C-a-FRET-ratio.png)
Stability of the Glu[−] sensor (20 µM) in MOPS buffer (20 mM, pH 7.3)... | Download Scientific Diagram