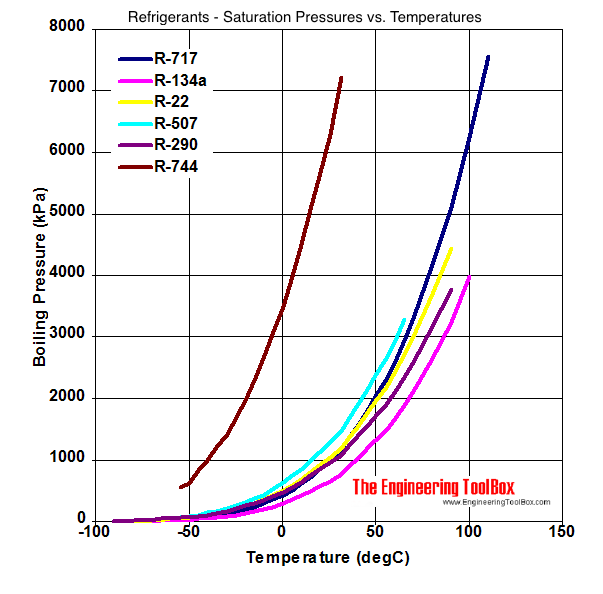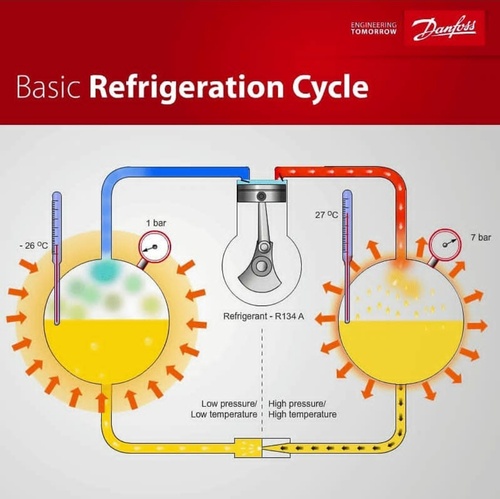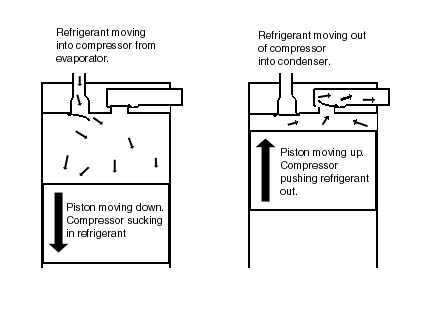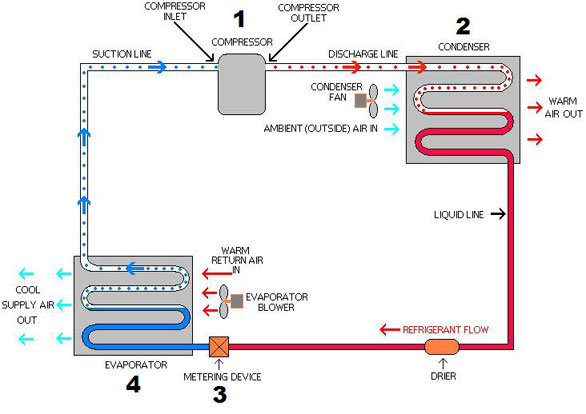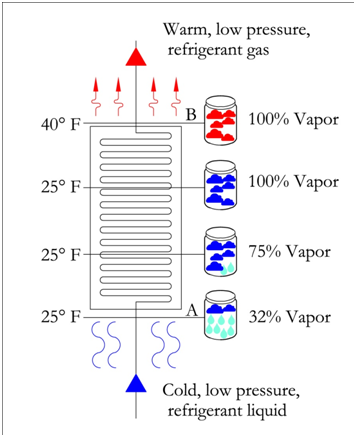
P-H Diagram Thermodynamics | HVAC and Refrigeration PE Exam Tools | Mechanical and Electrical PE Sample Exams, Technical Study Guides and Tools

QUIZ 2 A refrigerator uses refrigerant-134a as the working fluid and operates on an ideal vapor-compression refrigeration cycle between 0.18 and 0.9 MPa. - ppt video online download

Comparison of evaporation temperature vs. refrigeration duty for R22... | Download Scientific Diagram

SOLVED:The heat of vaporization of liquid Freon-12, CCl2 F2 is 4.71 kcal / mol. Calculate the energy required to vaporize 39.2 g of this compound. The molecular weight of Freon-12 is 120.9 amu.

SOLVED: The heat of vaporization of freon (CClaFz) is 34.4 kUlmol at 25 "€ What is the change of entropy for one mole of liquid freon when it vaporizes at 25 *c? (