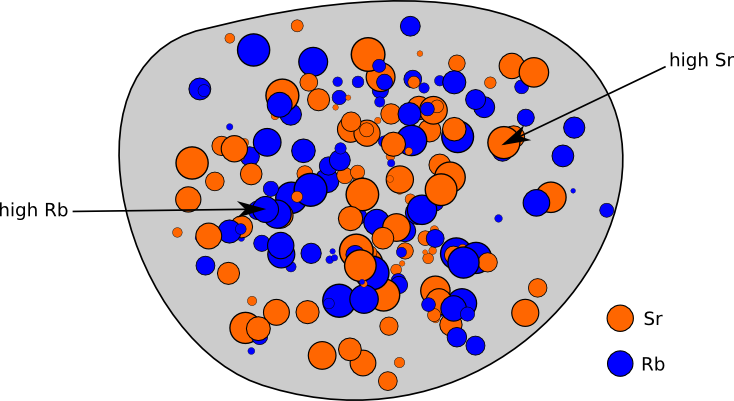
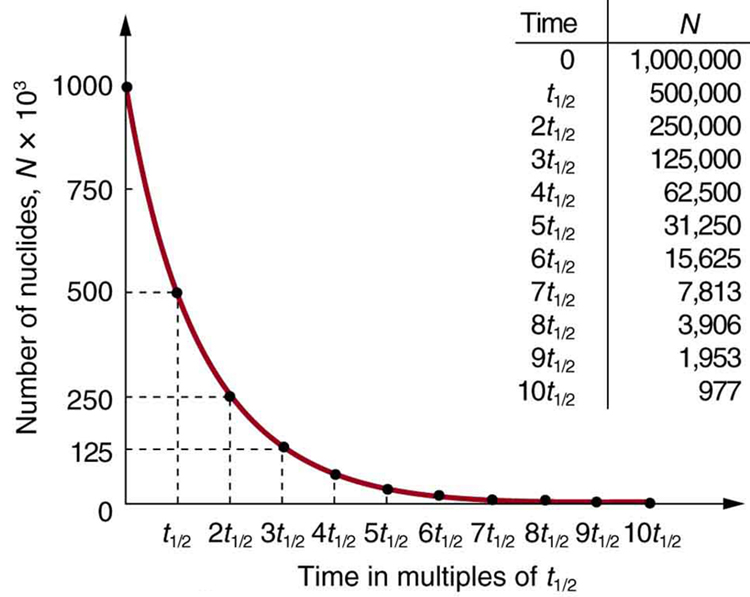
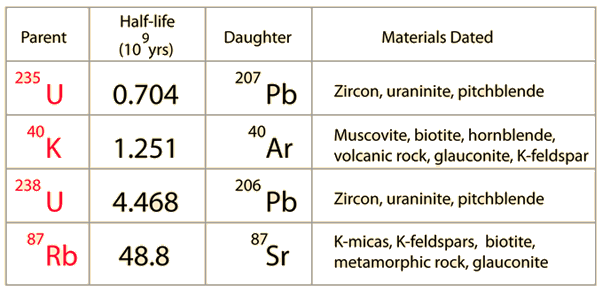
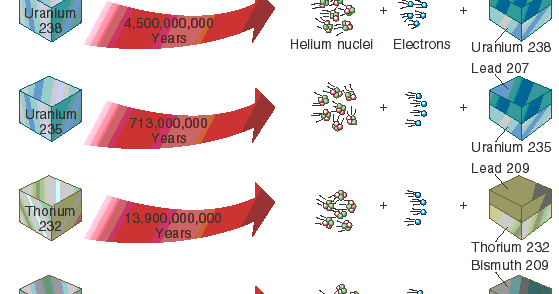
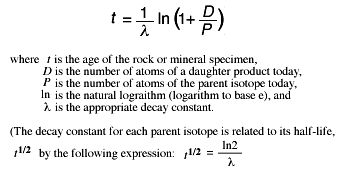21 - chemistry - 21: Radioactive Decay Recognize common modes of radioactive decay Identify common - Studocu

Radiometric Dating Methods & Examples | What is Radiometric Dating? - Video & Lesson Transcript | Study.com
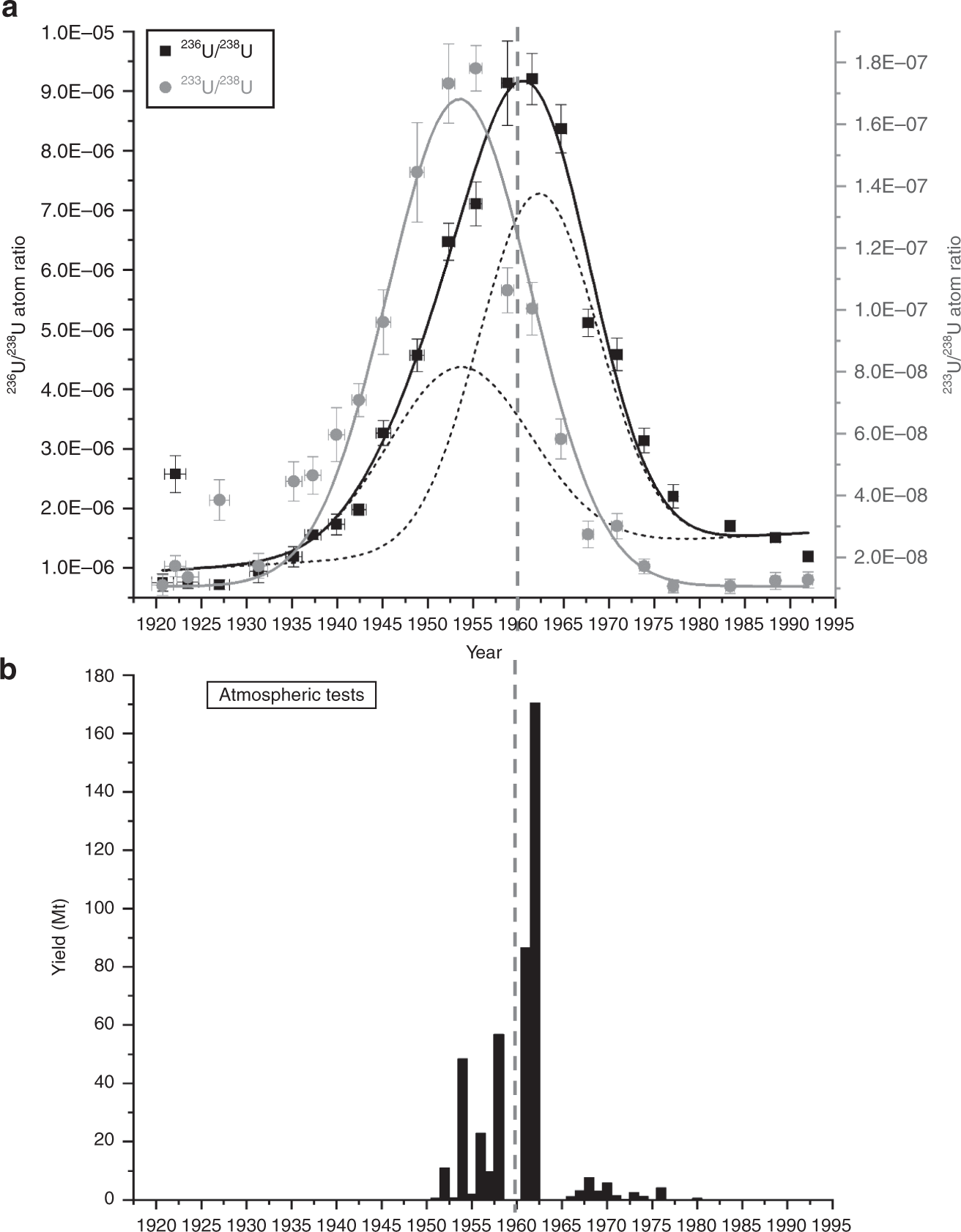
233U/236U signature allows to distinguish environmental emissions of civil nuclear industry from weapons fallout | Nature Communications

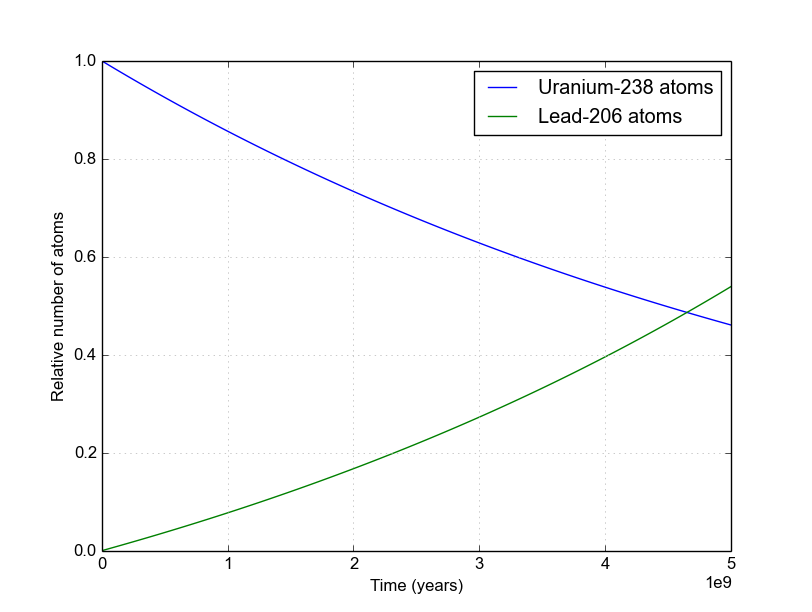
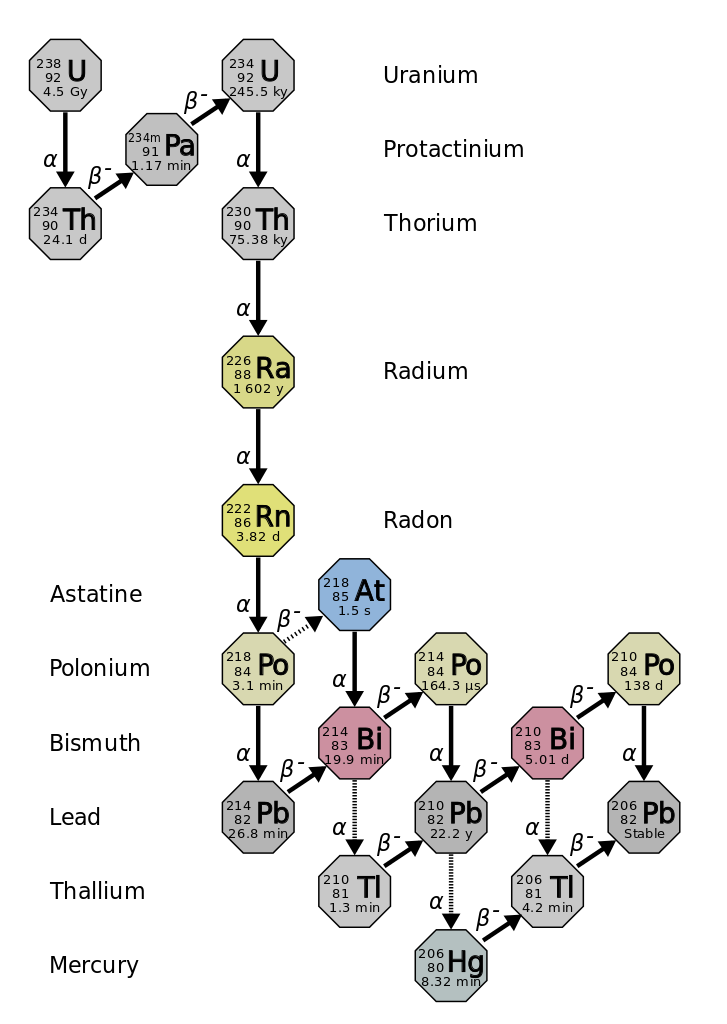
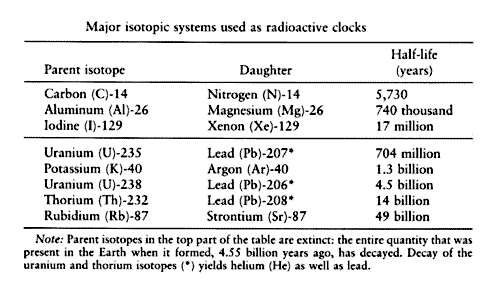
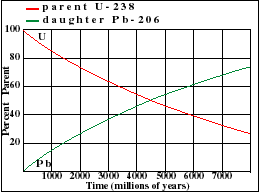
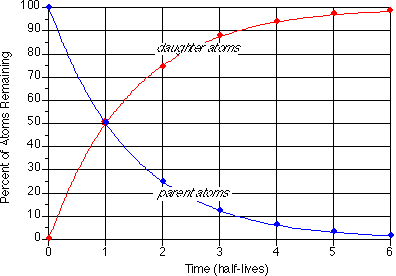
The final product of U^(238) is Pb^(206). A sample of pitchblende contains 0.0453 g of Pb^(206) for every gram of U^(238) present in it. Supposing that the mineral pitchblende formed at the

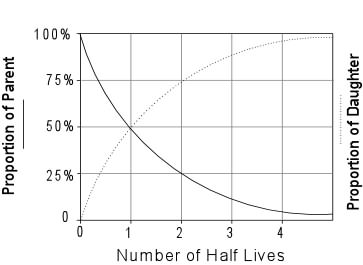


:max_bytes(150000):strip_icc()/concordia-58b59afb5f9b5860468034a4.jpg)