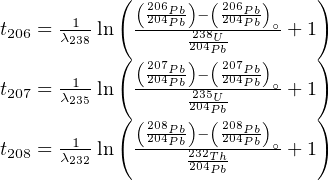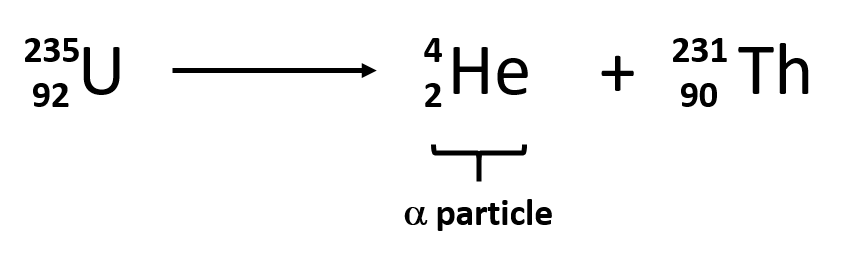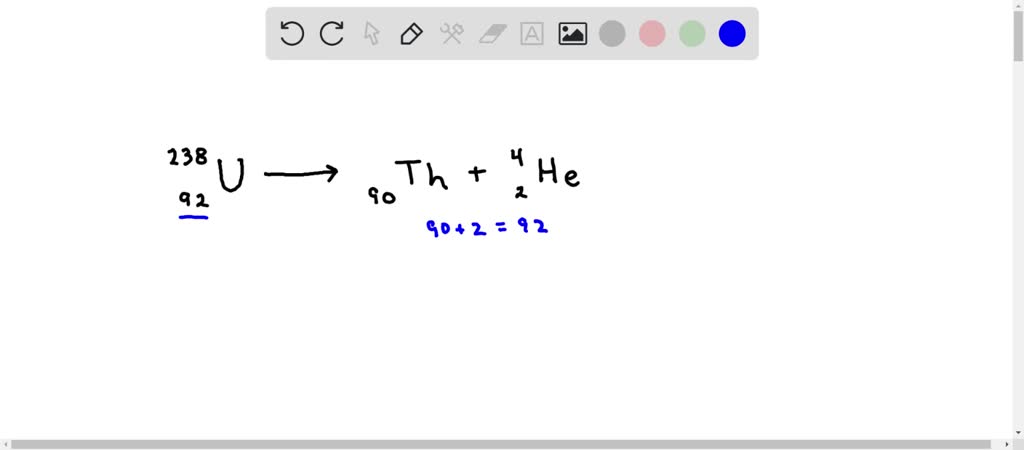
SOLVED: 21 Uranium-238 follows a first order radioactive decay to thorium-234 by the release of one alpha particle The nuclear equation that represents the process is: %8U 298Th {He Which of the
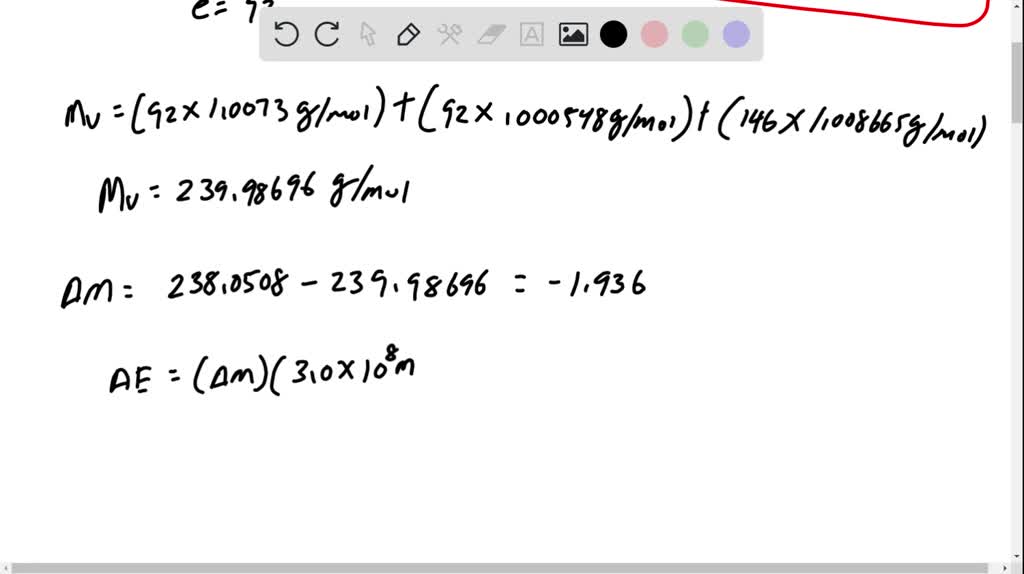
SOLVED:The most abundant isotope of uranium is ^238 U, which has an isotopic mass of 238.0508 g / mol. Calculate its nuclear binding energy in kJ / mol and its binding energy per nucleon.
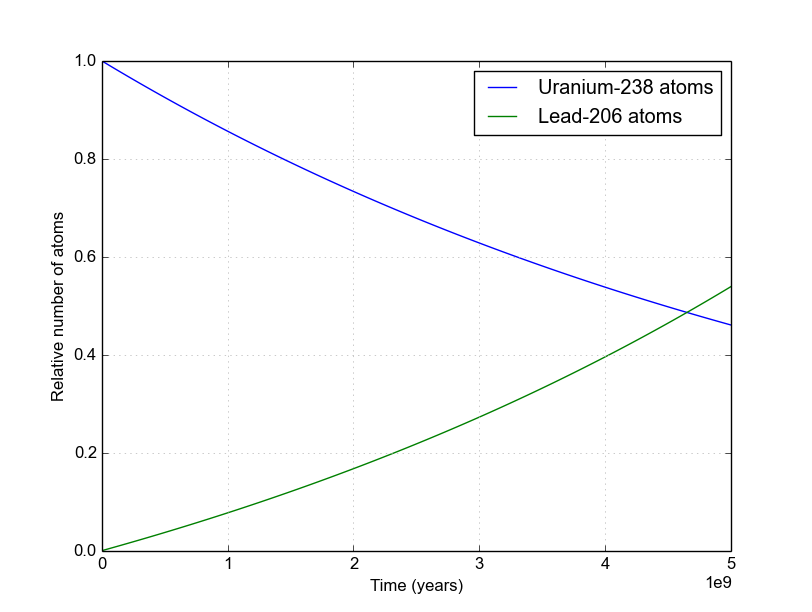
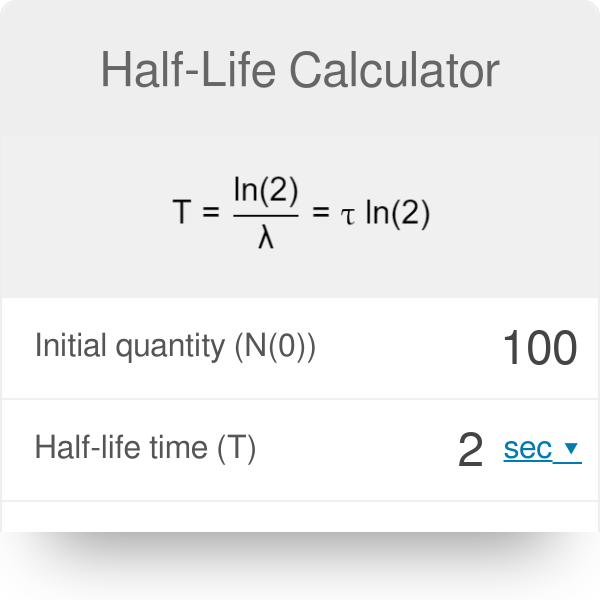
How is the precise rate of decay determined for atoms that have deep time half-lives, such as uranium-235 to lead-207? - Quora

OpenStax College Physics Solution, Chapter 31, Problem 57 (Problems & Exercises) | OpenStax College Physics Answers

The half - life of ^238 U for alpha - decay is 4.5 × 10^9 years. The number of disintegration per second occur in 1g of ^238 U is (Avogadro's number = 6.023 10 ^23 mol^-1 )


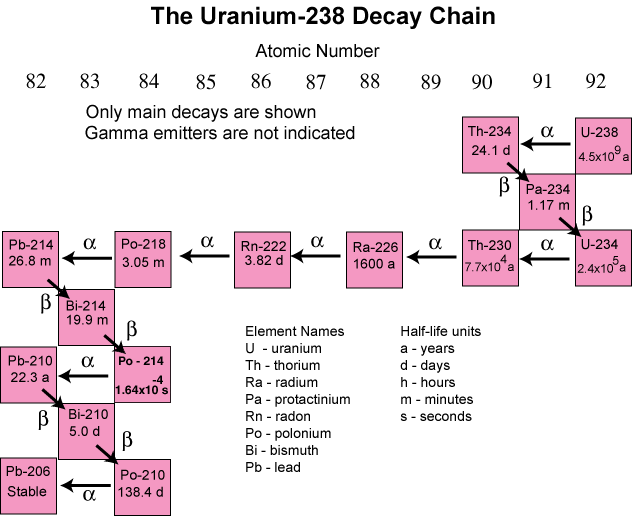
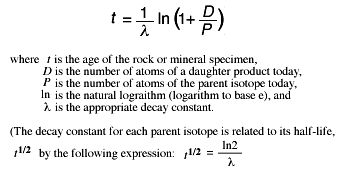

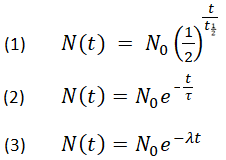

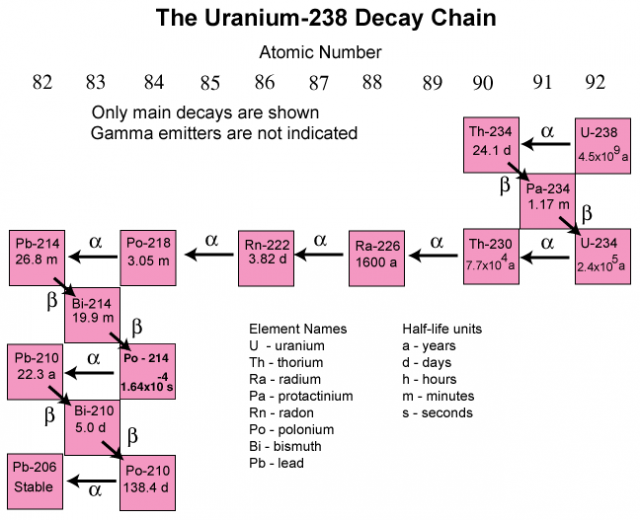
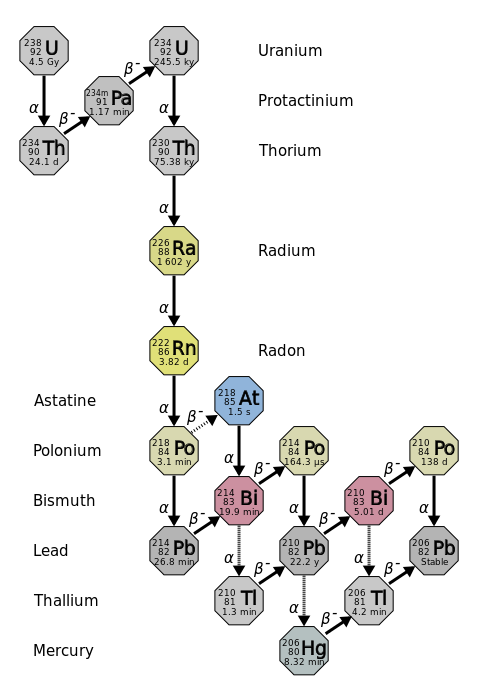.svg.png?revision=1)