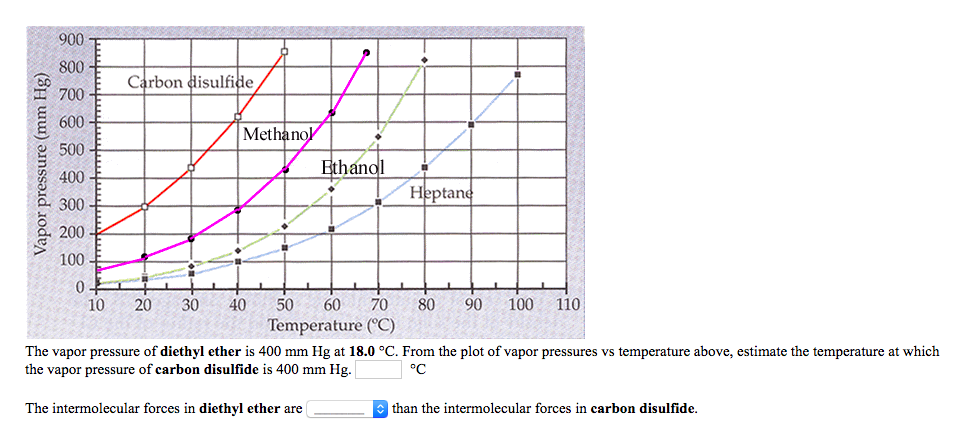
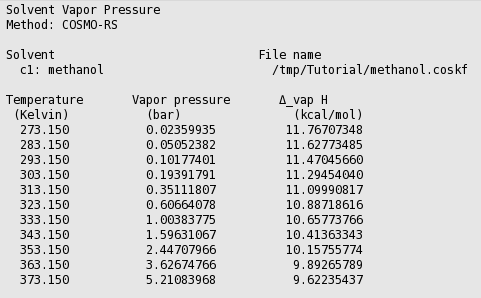
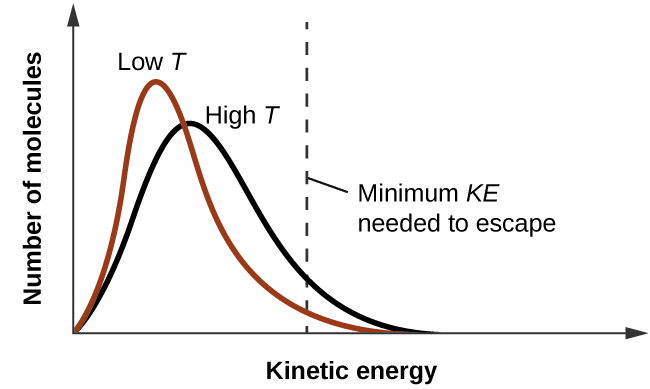
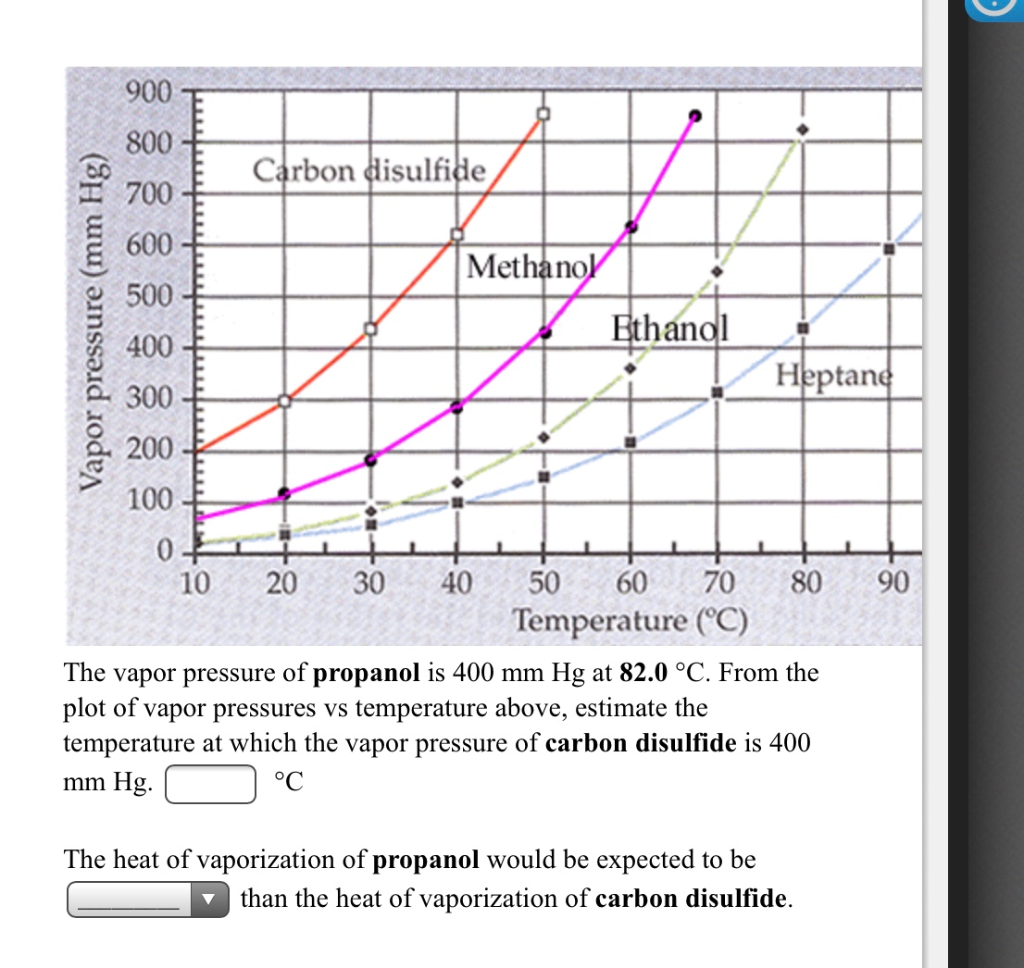
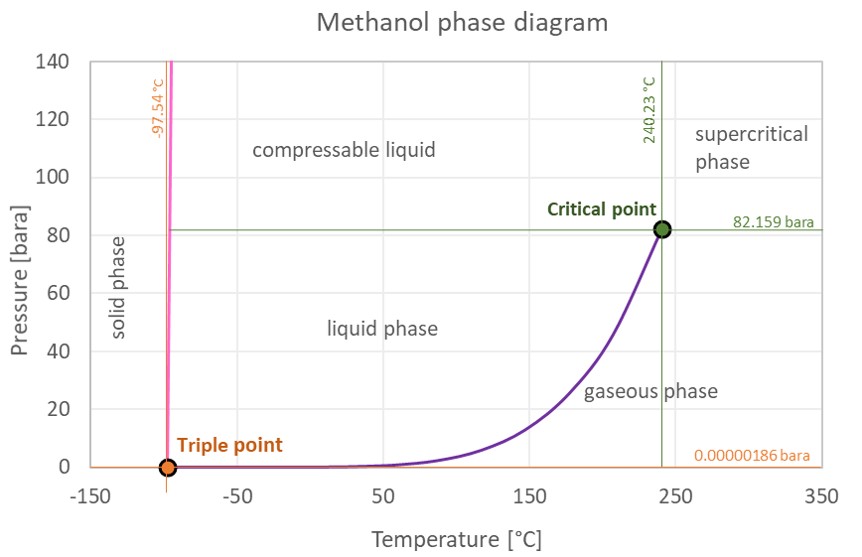
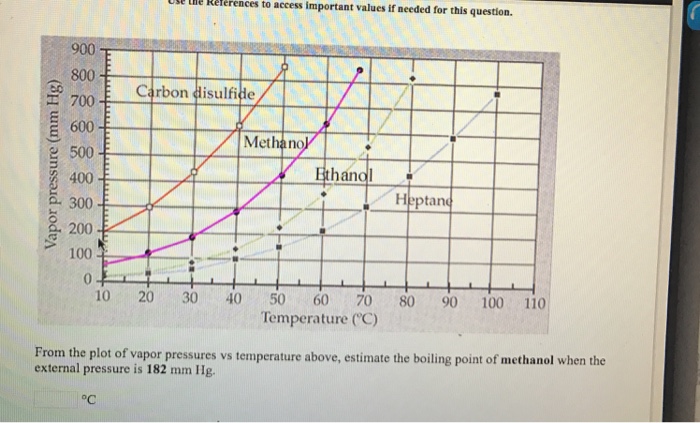
Solved] 900 800 Carbon disulfide 700 600 Methanol 500 Vapor pressure (mm Hg) Ethanol Heptane 300 200 100 10 20 30 40 50 60 70 80 90 100 110 Temperat... | Course Hero

The vapour pressure of ethanol and methanol ate `44.5 mm Hg` and `88.7 mm Hg`, respectively. - YouTube

OneClass: What is the vapor pressure at 20 degree C of an ideal solution prepared by the addition of ...

Vapor Pressures and Thermophysical Properties of Dimethoxymethane, 1,2-Dimethoxyethane, 2-Methoxyethanol, and 2-Ethoxyethanol: Data Reconciliation and Perturbed-Chain Statistical Associating Fluid Theory Modeling | Journal of Chemical & Engineering Data

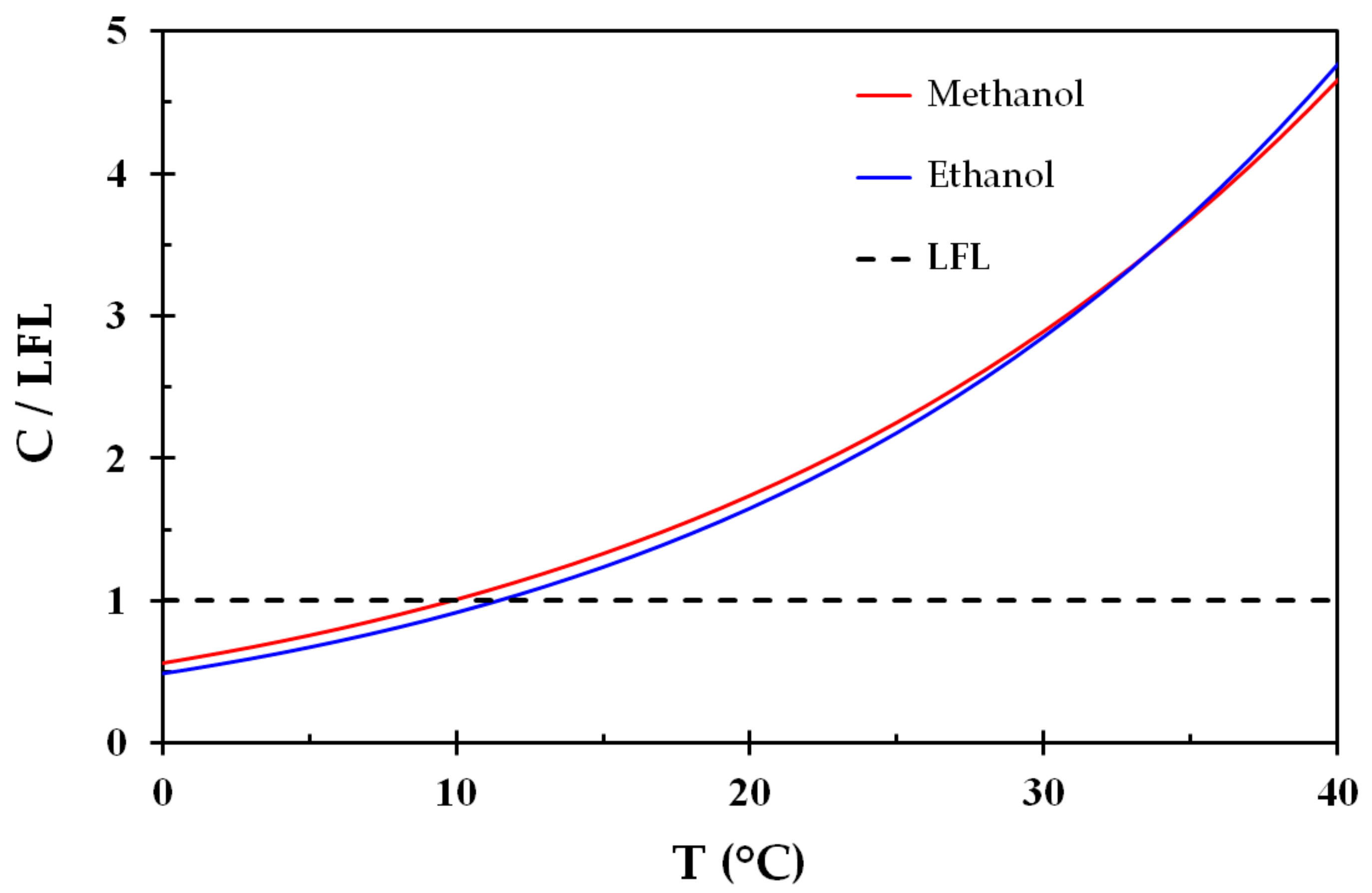
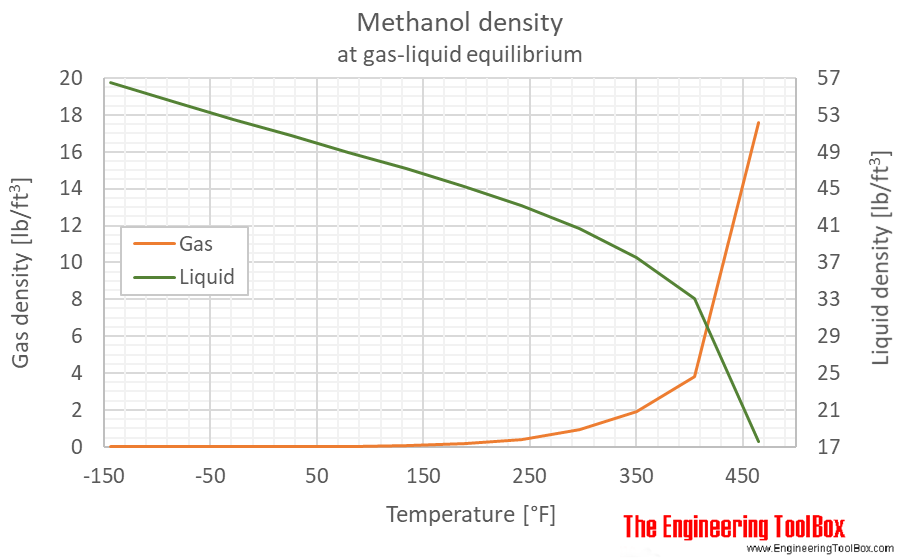
Vapor pressure of methanol and ethanol as a function of temperature... | Download Scientific Diagram

The vapour pressure of ethanol and methanol are 44.0 mm and 88.0 mm Hg, respectively. An ideal solution is formed at the same temperature by mixing 60 g of ethanol with 40g