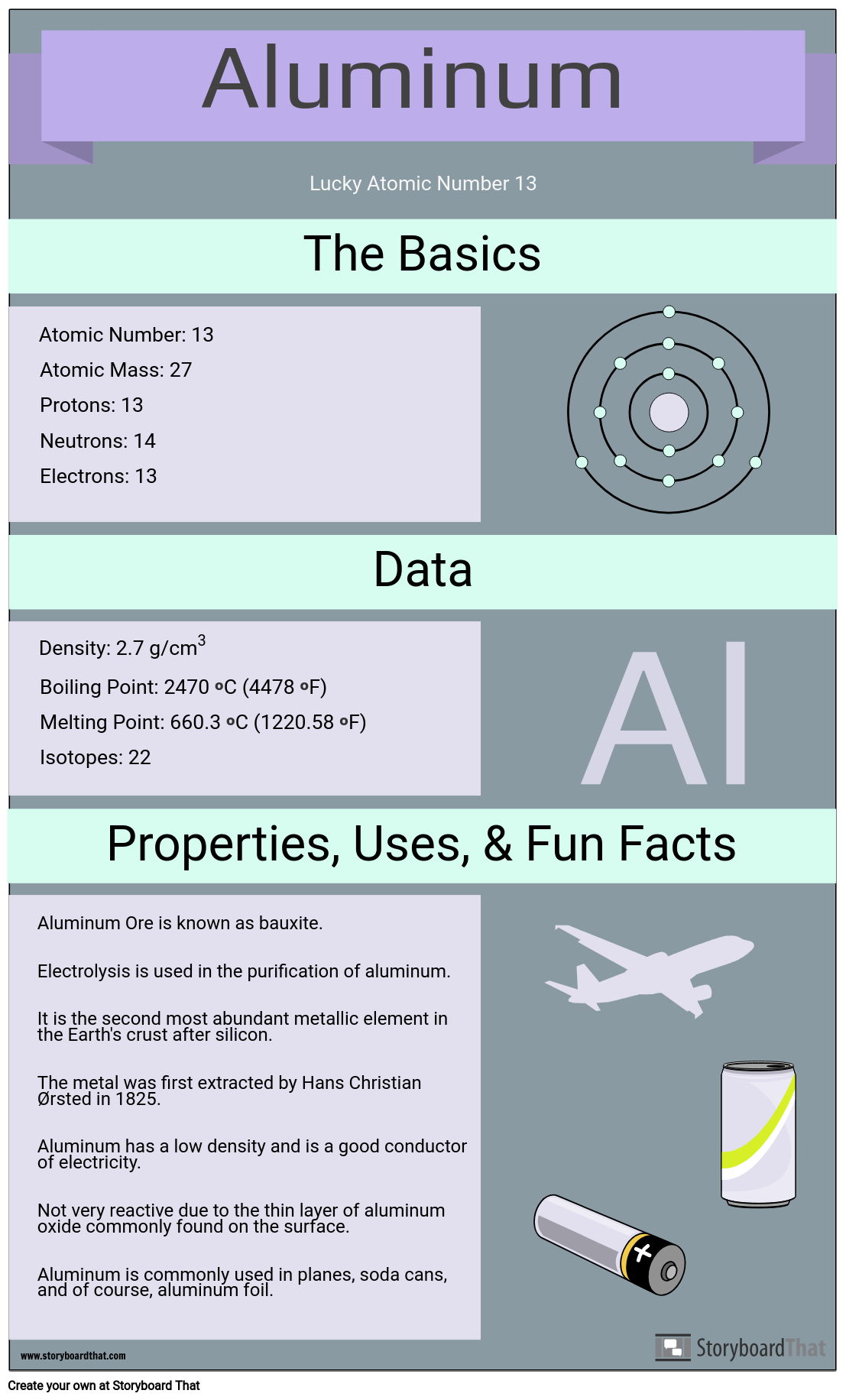
An element has atomic number 13 and an atomic mass of 27 (a) How many electrons are there in each atom of the

Aluminum Is A Chemical Element With The Symbol Al And Atomic Number 13 With A Mass Of 27 U Aluminum Stone For Industrial Use Silver Ore Stock Photo - Download Image Now - iStock

The Periodic Table: atom, chemistry, dmitri, dobereiner, element, eng, mendeleev, newlands, periodic, set | Glogster EDU - Interactive multimedia posters

Write down the characteristics of the elements having atomic number 17.Electronic configuration Period number Group number Element family No. of valence electrons Valency Metal or non - metal .

Aluminium symbol. Element number 13 of the Periodic Table of the Elements - Chemistry - Black square frame with white background Stock Vector Image & Art - Alamy

an element X has atomic mass 27 and atomic number 13 first draw the atomic structure of atomic X when - Brainly.in

SOLVED:A substance has 13 protons, 14 neulrons_ and 13 clectrons_ Which isotope is it? Al-13 B) Al-14 Al-27 D) Al-40 One isotope of oxygen has the atomic number 8 and the mass

1. what is the name of element, for this symbol Fe, the atomic number, mass number, protons,neutrons, electrons 2. what... - HomeworkLib

Element X has atomic number 13 and mass number 27 and element Y has atomic number 17 and mass number 35. (i) Write the electronic configuration of both X and Y (ii)

An element 'X' has atomic number 13:; (a) Write its electron configuration.; (b) state the group to - Brainly.in

An atom of an element contains 13 electrons. Its nucleus has 14 neutrons. Fid out its atomic number and approx

An element has atomic number 13. Which of the following statements concerning this element is wrong?(A) the atom has 13 protons(B) the element is placed in the third period of the periodic
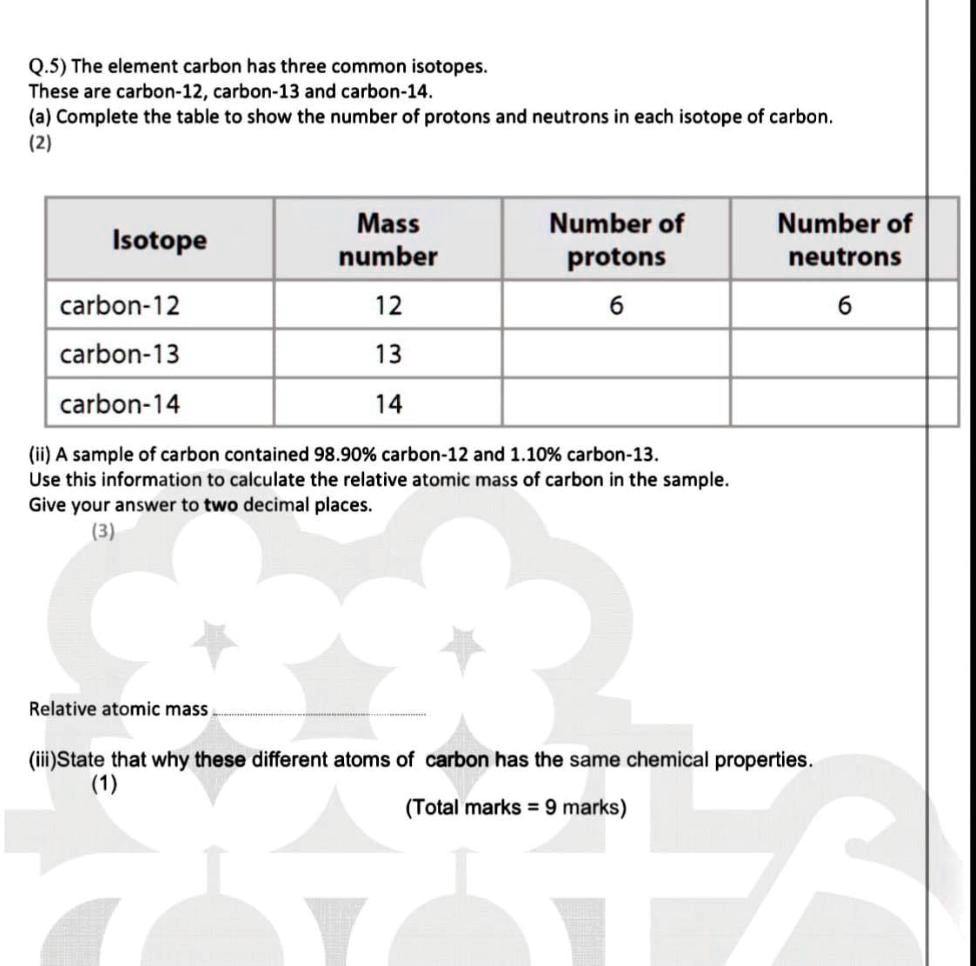
SOLVED:Q.5) The element carbon has three common isotopes: These are carbon-12, carbon-13 and carbon-14 (a) Complete the table to show the number of protons and neutrons in each isotope of carbon: (2)

/what-are-the-first-20-elements-608820-FINAL-5b758ab446e0fb002c67279a.png)